
QbD Group awarded Platinum label as Best Managed Company 2026
QbD Group awarded Platinum label as Best Managed Company 2026, marking seven consecutive years of recognition for excellence in the life sciences industry.
阅读更多
QbD Group awarded Platinum label as Best Managed Company 2026, marking seven consecutive years of recognition for excellence in the life sciences industry.
阅读更多
As pharmaceutical organizations grow and evolve, Lifecycle Management becomes increasingly difficult to control. This webinar explores how complexity develops across Regulatory Affairs and Pharmacovigilance, and how to structure activities in a more scalable and cost-efficient way.
阅读更多
At QbD Group, we are joining forces with Vigintake to support the evolution of pharmacovigilance operations through intelligent automation and innovative digital solutions, combining regulatory expertise, quality systems, and advanced technology.
阅读更多
QbD Group today announces the acquisition of Rescop, a specialist in GxP compliance, computer system validation, and validation software for the life sciences industry.
阅读更多
QbD Group has been recognized with the AEFI Award for Best Publication of 2025 for the article "Pharmacovigilance in innovative therapies: Regulatory and clinical challenges," authored by Almudena del Castillo, Head of Global Vigilance Division and Country Manager Spain.
阅读更多
Successful MedTech companies treat product development as a lifecycle strategy. Learn why regulatory planning, IP strategy and development must evolve together.
阅读更多
Leveraging AI in life sciences is powerful, but also complex under GxP. In this joint webinar, QbD Group and delaware show how to implement AI in a trustworthy, validated, and inspection-ready way, with the right approach to data, explainability, and lifecycle management.
阅读更多
Early-stage biotech teams often struggle to translate scientific innovation into first-in-human readiness. This webinar provides practical guidance to help teams build a clear, scalable development pathway that aligns regulatory strategy, CMC, quality, and clinical planning. Reducing risk, avoiding delays, and staying inspection-ready without overbuilding internal structures.
阅读更多
ICH E2D(R1) introduces new expectations for ODCS, PSPs, MRPs and digital platforms. Learn what MAHs must implement by 18 March 2026 to ensure pharmacovigilance compliance.
阅读更多
Manage the risk of nitrosamines in the pharmaceutical industry. Watch our webinar on demand and learn about regulation and risk analysis from experts.
阅读更多
This webinar follows the stages of a product’s life cycle, explaining how structured, science-based development and intellectual property considerations support regulatory alignment and long-term value creation.
阅读更多
Planning EU and UK expansion as a US biotech? Learn which regulatory, clinical and operational decisions matter from day one.
阅读更多
Residual solvent testing is essential to patient safety and regulatory compliance. Learn how GC-FID and GC-MS support ICH Q3C compliance in pharmaceutical manufacturing.
阅读更多
As European SMEs and biotechs expand, PV governance must scale to maintain EU-QPPV oversight and inspection readiness. Learn how to build a scalable pharmacovigilance system.
阅读更多
Learn how Annex 1 expectations for cleanrooms, isolators and RABS shape modern aseptic manufacturing. Practical guidance on barrier strategy, airflow visualization and contamination control.
阅读更多
Orphan strategy decisions go beyond incentives. Learn how regulatory definitions, evidence requirements, timing, and exclusivity across the EU, US, and UK shape early biotech development strategy.
阅读更多
Life sciences companies must digitalize fast while staying GxP compliant. In this joint webinar, QbD Group and delaware show how SAP technology and proven validation frameworks make rapid, compliant digital transformation possible. Participants will learn best practices for SAP implementation under GxP, how to avoid common pitfalls in validation and CSV, and gain insights into joint validation methodologies and real-world examples where innovation meets compliance.
阅读更多
On 01/12/2025, the European Medicines Agency (EMA) released an update to Appendix 1 (EMA/42261/2025/Rev. 11) of the Nitrosamines Guideline. This appendix lists nitrosamines for which an established Acceptable Intake (AI) is available.
阅读更多
Learn the key requirements for validating Single-Use Systems, with guidance on supplier control, compliance, and Extractables & Leachables risk management.
阅读更多
QbD Group & delaware announce a new partnership to help life sciences companies accelerate their digital transformation while ensuring full GxP compliance.
阅读更多
Ph. Eur. Chapter 2.4.35 defines a harmonized approach to assess extractable elements in plastic pharmaceutical materials, strengthening risk-based control of elemental impurities.
阅读更多
This webinar provides guidance on managing pharmacovigilance (PV) across Europe, focusing on balancing global consistency with country-specific requirements. Participants will learn to meet strict European timelines while working with lean teams and practical strategies to enhance oversight and execution.
阅读更多
QbD Group joins ASEBIO, strengthening its commitment to quality, compliance, and safety within the Spanish life sciences sector.
阅读更多
Learn what the new EudraLex Annex 22 means for AI regulation in pharma and how it could impact your daily operations and compliance efforts.
阅读更多
ICH Q2(R2) and Q14 redefine method validation as a continuous lifecycle. Learn how to keep analytical methods fit for purpose and inspection-ready.
阅读更多
Global PV success requires more than compliance. Discover how hybrid pharmacovigilance models help pharma companies meet local regulatory demands and build patient trust.
阅读更多
FDA is expanding unannounced inspections at foreign sites. Learn how manufacturers can stay inspection ready through resilient systems, validated processes, and confident people.
阅读更多
Cybersecurity is a key MDR requirement. Discover what your technical file must include to meet EU cybersecurity expectations for medical devices.
阅读更多
The EU is revising Annex 11 to align GMP with digital realities. Discover what’s changing and how to prepare for compliance before 2026.
阅读更多
Discover why effective project governance is key to global pharmacovigilance. Ensure compliance, transparency, and patient safety across markets.
阅读更多
QbD Group has been awarded the title of Best Pharmacovigilance Service Company at the latest edition of Farmaforum.
阅读更多
The European Commission has amended Regulation (EU) No 520/2012 on pharmacovigilance. The update strengthens oversight of outsourced PV activities, clarifies EudraVigilance monitoring, and aligns processes with international data standards (IDMP, MedDRA, Standard Terms).
阅读更多
Learn how Commission Implementing Regulation (EU) 2025/1234 broadens electronic Instructions for Use (eIFUs) for professional medical devices, the compliance steps required, and how IFUcare can help.
阅读更多
Learn how GMP labs can strategically balance quality, cost, and time using smart technologies, risk-based approaches, and lean workflows.
阅读更多
Learn how IVDR aligns with FDA standards, where they diverge, and how to use your IVDR compliance as a springboard for global IVD market access.
阅读更多
QbD Group welcomes Dr. Diane Fairweather as Division Head Clinical, with 25+ years in global clinical leadership to strengthen and scale the division.
阅读更多
QbD Group awarded Gold Label as Best Managed Company 2025, marking six consecutive years of recognition for excellence in the life sciences industry.
阅读更多
Learn how to optimize pharmacovigilance systems and outsource strategically to turn compliance into business growth.
阅读更多
Learn how Good Distribution Practices (GDP) and Quality Risk Management (QRM) ensure the integrity, safety, & compliance of pharma products during distribution.
阅读更多
The webinar discusses the evolving pharmacovigilance and risk management environment of innovative therapies, emphasizing safety challenges in cell, CAR T, and RNA-based treatments, as well as gaps in regulation and surveillance strategies. Participants will gain insight into the origin and impact of safety concerns in these therapies, alongside ongoing developments in RNA-based treatments.
阅读更多
This webinar offers a foundational understanding of Extractables and Leachables (E&L), highlighting their impact on drug safety and stability, and current regulatory frameworks. It covers toxicological assessments and best practices in study design, supported by real-world examples to navigate the complex landscape surrounding E&L.
阅读更多
Learn how to streamline your eCTD publishing and submissions with 10 expert tips. Avoid pitfalls, ensure compliance, and meet global regulatory requirements.
阅读更多
Dr. Angeles Escarti-Nebot joins QbD Group as Head of Regulatory Affairs, bringing global regulatory expertise to advance innovation in life sciences.
阅读更多
Prepare a compliant IMPD for clinical trials. Avoid delays with expert tips on quality documentation, common pitfalls, and regulatory expectations.
阅读更多
Veterinary pharmacovigilance audits are key to global health. Discover how QbD helps you comply with VGVP and ensure veterinary drug safety.
阅读更多
Learn the essentials of Pharmacovigilance Agreements (PVA) and Safety Data Exchange Agreements (SDEA) to ensure regulatory compliance and effective collaboration.
阅读更多
Explore how pharma companies can make software compliance a value-adding asset, enhancing efficiency, data security, and regulatory adherence.
阅读更多
Discover a comprehensive approach to maintaining software compliance in pharma. Explore compliance frameworks, key stakeholder roles, task management, and best practices.
阅读更多
Explore essential steps to maintain compliance in pharmaceutical software, from periodic reviews to change control, ensuring data integrity and safety.
阅读更多
Azierta integrates into QbD Group, expanding services and global reach, enhancing client value and employee opportunities in life sciences consultancy.
阅读更多
QbD Group unveils a new brand identity to reflect its growth, expanded services, and commitment to innovation in the life sciences sector.
阅读更多
Almudena del Castillo joins QbD Group as Division Head of Vigilance and Country Manager for Spain, strengthening leadership and patient safety.
阅读更多
Join our webinar to explore the essential clinical evidence needed for success, including investigations, literature reviews, technical performance, and the use of similar devices. We’ll discuss the critical role of clinical validation starting from the design phase and guide you on how to compile the necessary evidence to meet MDR requirements.
阅读更多
This webinar will explore the specific requirements for various safety classifications and provide valuable insights into what Notified Bodies (NBs) and the FDA expect from your development process. Key topics will include the full development cycle, cybersecurity essentials, usability engineering, AI integration, and effective strategies for aligning agile methodologies with the strict release flows required for MDSW. Gain a deeper understanding and streamline your MDSW development process today!
阅读更多
This webinar will guide you through the essential steps of market access, effective commercialization strategies, and all the critical factors needed for a successful MDSW product launch. Whether you’re a seasoned professional or new to the field, this session will provide you with the knowledge and tools to make your MDSW vision a reality.
阅读更多
Explore the regulatory complexities of Drug-Device Combination Products. Learn about Article 117 requirements, Notified Body Opinions, and more.
阅读更多
This webinar aims to explore the transformation of safety teams from reactive cost centers to proactive benefit drivers, emphasizing the importance of providing teams with the right tools & resources to gain actionable insights.
阅读更多
QbD Group and SciencePharma have reached an agreement for QbD Group to acquire SciencePharma.
阅读更多
Watch our webinar to gain practical insights on crafting essential technical documentation (TD) with confidence. We’ll take you step-by-step through interpreting MDR requirements, choosing the right accredited Notified Body, and navigating the qualification and classification process—all with a focus on your software’s Intended Purpose. Don’t miss this opportunity to ensure your documentation is not just compliant, but also clear and comprehensive.
阅读更多
Watch our webinar where we address major challenges in software qualification and classification for different functionalities. We explore MDR for software-hardware combinations and how AI is handled under the AI Act. In addition, we share essential resources, key standards, and guidance documents crucial for getting your product to market, from design and development to clinical evidence and technical documentation.
阅读更多
This webinar provides invaluable information on how to address and manage toxicological risks in the pharmaceutical industry, emphasizing the importance of timely and transparent communication. The session will cover essential strategies for assessing and interpreting risks, reporting findings, investigating root causes, and implementing corrective measures to ensure patient and environmental safety.
阅读更多
Learn how to design and deliver successful clinical performance studies under IVDR. Explore strategic, regulatory, and operational aspects with expert guidance.
阅读更多
During this webinar you will learn how to effectively prepare for a compliant ATMP Quality Management System. Discover the key elements required to establish and maintain a QMS that adheres to Eudralex Part IV regulations.
阅读更多
In this webinar we dive into the fascinating world of the ATMP (Advanced Therapy Medicinal Products) supply chain. Discover how to overcome the main challenges faced by industry leaders and gain fascinating insights into the innovative vein-to-vein strategy. Through real-life examples and visuals, we will explore the roadmap to navigate the ATMP supply chain and unlock its full potential.
阅读更多
This webinar will tell you more about the second edition of GAMP 5, which will provide updates, clarify the relationship between GAMP 5 and the GAMP® 5: Records and Data Integrity Guide, recognition of the FDA’s current work on computer software assurance (CSA), and an updated, dynamic and evolving set of Appendices.
阅读更多
Learn how technovigilance ensures medical device safety through post-market surveillance, tackling risks, compliance, and best practices.
阅读更多
Discover how combining unscripted and scripted testing ensures robust Computer System Validation (CSV) for compliance, accuracy, and system reliability.
阅读更多
The webinar provides an exclusive insight into healthcare manufacturing 3D printing. Learn from the direct expertise of Michaël Vandezande, Stijn Paridaens and Anne-Sophie Grell. With their vast knowledge in prototyping as well as in mass-manufacturing via 3D printing, they will explore the challenges, opportunities, and regulations of additive manufacturing in the Healthcare sector.
阅读更多
The journey towards adopting the IVD regulations introduced in 2017 has been ongoing for some time, presenting challenges for many in the industry. This session will provide you with the latest updates on the IVDR extension and its impact on your daily operations and long-term strategies. Don’t miss this opportunity to gain valuable insights directly from an expert at the forefront of the field.
阅读更多
Manage yearly audits to ensure compliance and quality in pharma and medical devices. Learn key strategies and boost your QMS now!
阅读更多
Annual update on the QbD Research Chair for Precision Oncology in glioblastoma, highlighting patient-centered care, research progress, and a tribute.
阅读更多
Achieving inspection readiness ensures you're always prepared for regulatory inspections. Learn how to stay compliant and confident every day.
阅读更多
QbD Group earns its fifth consecutive Gold label as Best Managed Company, recognized for its exceptional strategy, competencies, and financial results.
阅读更多
Discover what happens during a regulatory inspection, what to expect from start to finish, and how to prepare for a smooth, stress-free inspection process.
阅读更多
Explore the FDA's final rule integrating ISO 13485:2016 into QMSR for medical devices. Understand impacts on manufacturers and how to comply by 2026.
阅读更多
Gain an insightful understanding into how the MHRA inspect the Pharmacovigilance services of an MAH for post-authorised products.
阅读更多
In this blog post, we delve into the intricacies of Digital Therapeutics (DTx) regulation in the UK, especially in light of Brexit in January 2020.
阅读更多
Discover how to create a GMP-compliant stability protocol for pharma, ensuring product safety and efficacy throughout its shelf life.
阅读更多
The US has a solid regulatory framework for Digital Therapeutics, requiring strong clinical evidence and robust data protection measures. Learn more here.
阅读更多
When it comes to the regulation of Digital Therapeutics (DTx), Germany is pioneering new paths for its adoption. This blog post delves into the intricacies of Germany's approach to DTx regulation.
阅读更多
In this blog post, we delve into the DTx regulations of Belgium and France, focusing on their reimbursement strategies and providing a glimpse into how other EU member states fare in comparison.
阅读更多
Timeseer.AI and QbD Group enhance sensor data quality in life sciences, ensuring reliable information and better patient care.
阅读更多
QbD Chair in Precision Oncology for Glioblastoma is extended three years, honoring a personal story and advancing innovative cancer treatments.
阅读更多
Salma Michor joins QbD Group as Head of Regulatory Affairs, bringing extensive expertise and leadership to strengthen the regulatory team.
阅读更多
Bart Van Acker honors a friend's memory by funding research on glioblastoma, aiming to accelerate treatment development through a new chair at Leuven University.
阅读更多
Salma Michor honored as a 2023 RAPS Fellow for her exceptional leadership and contributions to the regulatory profession.
阅读更多
Watch our two on-demand webinars and discover how to turn PMS & PMCF compliance into clinical and strategic advantage — with exclusive insights from a notified body.
阅读更多
In this webinar on demand, we’ll reveal how to transform technical information into a compelling website that connects with your audience. This session will address critical questions that impact your organization’s reach and reputation. You will walk away with practical strategies for creating content that conveys your scientific expertise while also being accessible and engaging. Whether you’re launching a new product or seeking to strengthen your brand’s online presence, this webinar will equip you with insights and tools to communicate effectively and make a lasting impact in the life sciences market.
阅读更多
This webinar on demand will explain how PMS includes all the activities carried out by the manufacturer to monitor, measure, and assess the device’s quality, performance and safety to ensure that the benefit-risk profile of the device remains current. We will also briefly dive into the concept of Post Market Clinical Follow-up (PMCF) for medical devices and Post-Market Performance Follow-up (PMPF) for IVDs.
阅读更多
Explore the essentials of plasmid manufacturing and regulations for applications like gene therapy and vaccine development.
阅读更多
Oncolytic viruses and cellular immunotherapy show promise in cancer care. Learn their mechanisms, risks, and key biosafety requirements.
阅读更多
QbD Group acquires Azierta to strengthen global vigilance services and expand its presence in Spain.
阅读更多
Belgian researchers launch a 3D bioprinted miniature heart to the ISS to advance understanding of heart aging and improve cardiovascular health.
阅读更多
Discover the future of pharma with paperless validation. Boost efficiency, cut costs, and drive agility and innovation in the digital era.
阅读更多
In this webinar on demand, we will provide a concise explanation of the process involved in qualifying and classifying a medical device. We will also discuss the most suitable method of conformity assessment to follow and shed light on how a Notified Body conducts audits of your Quality Management System (QMS) and Technical documentation.
阅读更多
Explore the key factors affecting medicinal product stability and the parameters to consider for proper storage and quality maintenance.
阅读更多
In this blog post, we will delve deeper into the significance of stability testing in pharmaceutical development.
阅读更多
Discover key changes in UK and EU clinical trial approvals, including the UK's Combined Review system and the EU Clinical Trials Regulation.
阅读更多
QbD Group acquires Michor Consulting to enhance regulatory affairs expertise and expand services in Europe, particularly in the DACH region.
阅读更多
QbD Group receives its fourth consecutive Best Managed Companies label and the Gold Label from Deloitte.
阅读更多
Discover ILAP: UK's Innovative Licensing and Access Pathway for faster patient access to advanced treatments through streamlined licensing.
阅读更多
Learn how the QbD Group Foundation strengthens social impact through science, healthcare, environmental initiatives, and local community engagement.
阅读更多
Are you a manufacturer of custom-made medical devices and in need of regulatory guidance? This post summarizes key concerns to consider.
阅读更多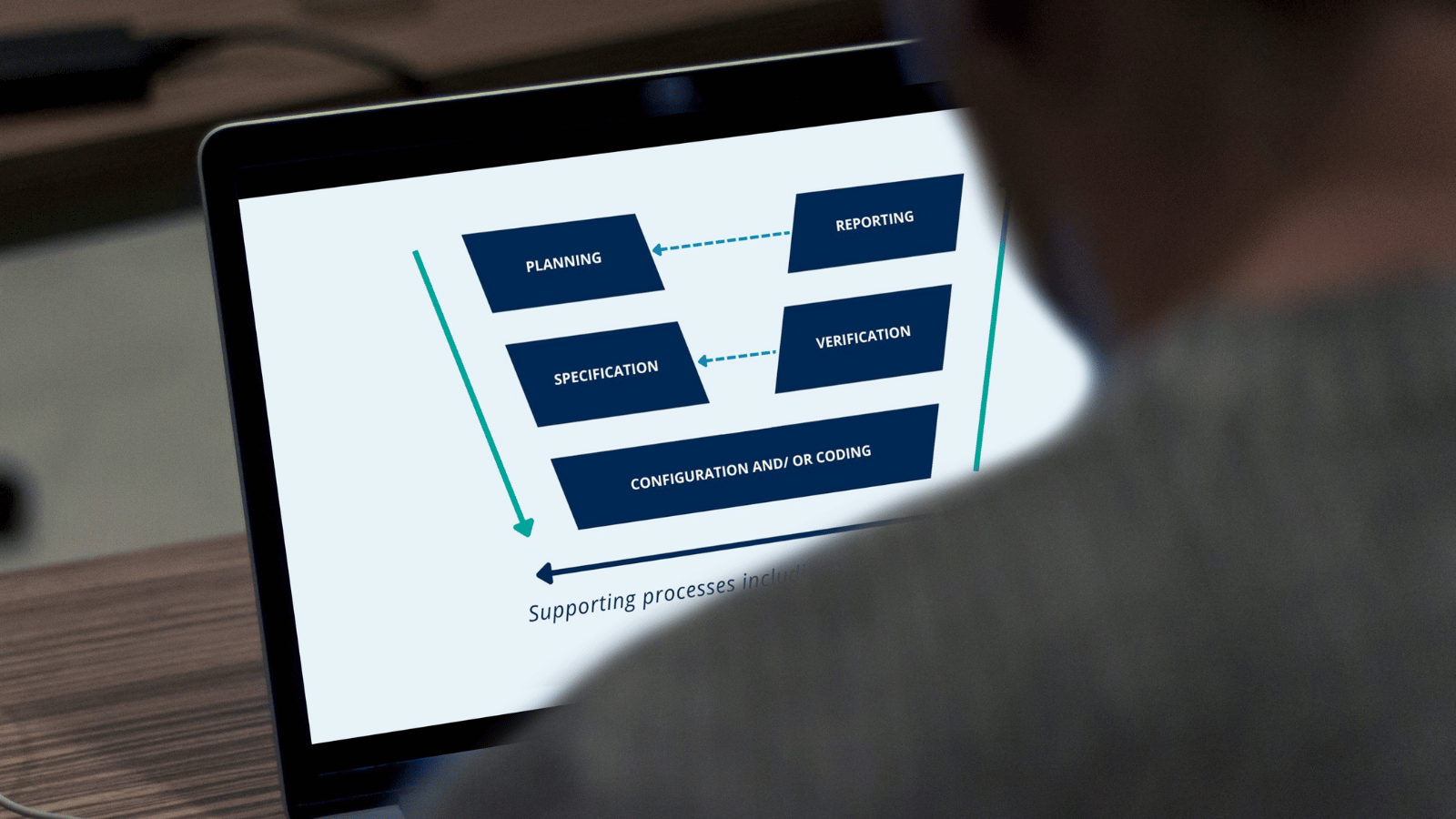
Want to introduce robustness into your software validation activities? In this article, you will learn what the GAMP 5 V-model is and the key steps within it.
阅读更多
Performing clinical research with medical devices? Learn more about all essential documents required during a medical device clinical investigation.
阅读更多
Each medical device needs a Unique Device Identifier (UDI) with a barcode or QR code. But what about medical device software? Find out in this article!
阅读更多
Discover whether pharma's computerized system validation applies to non-product medical device software by comparing ISO 80002-2 with GAMP5.
阅读更多
QbD Group acquires EUDRAC Ltd., expanding regulatory affairs and pharmacovigilance services.
阅读更多
Qarad rebrands its eIFU services as IFUcare, enhancing visibility and compliance for distributing medical device instructions digitally.
阅读更多
Digital health is on the rise, but what is digital health exactly? In this blog post, we'll explore the landscape, key technology pillars and future opportunities.
阅读更多
Quality by Design and TRIUM form a strategic alliance to offer comprehensive end-to-end solutions for the life cycle of medical products and therapies.
阅读更多
TRIUM Clinical Consulting has rebranded to QbD Clinical, expanding its service offerings within the QbD Group.
阅读更多
In CSV, decommissioning is the retirement or controlled shutdown of a computer system that is no longer needed. Learn more about this process.
阅读更多
QbD Wallonia expands with a new office in Gosselies, focusing on biotech and ATMP, and strengthens its team to drive growth.
阅读更多
Learn more about the new risk-based approach to in-vitro diagnostic device classification under IVDR 2017/746, replacing outdated directives.
阅读更多
Quality by Design expands its services by welcoming Qarad, enhancing its expertise in Regulatory Affairs and Quality Assurance for IVD.
阅读更多
The terms SaMD (Software as a Medical Device) and MDSW (Medical Device SoftWare) are often used interchangeably within medical environments, but they are not. Learn the difference in meaning and regulatory scope here.
阅读更多
Is your product a medical device? Well, that depends on the markets you want to enter and their regulations. Here's an overview to guide you.
阅读更多
Discover Scilife: QbD Software's rebrand to enhance value, user-friendliness, and industry leadership in Life Sciences.
阅读更多
QUERCUS Labo joins QbD Group, strengthening quality control services and expertise in life sciences.
阅读更多
Regulatory Affairs plays a key role in the pharmaceutical industry: from drug development to commercialization. Learn more about the roles and functions that RA teams can provide in the lifecycle management of your pharmaceutical product.
阅读更多