Operational software compliance in practice
Process and stakeholders in the operational phase
Compliance during the operational phase requires the involvement of multiple stakeholders, each with specific roles in ensuring regulatory adherence and risk mitigation. Below is a structured process to execute compliance tasks:
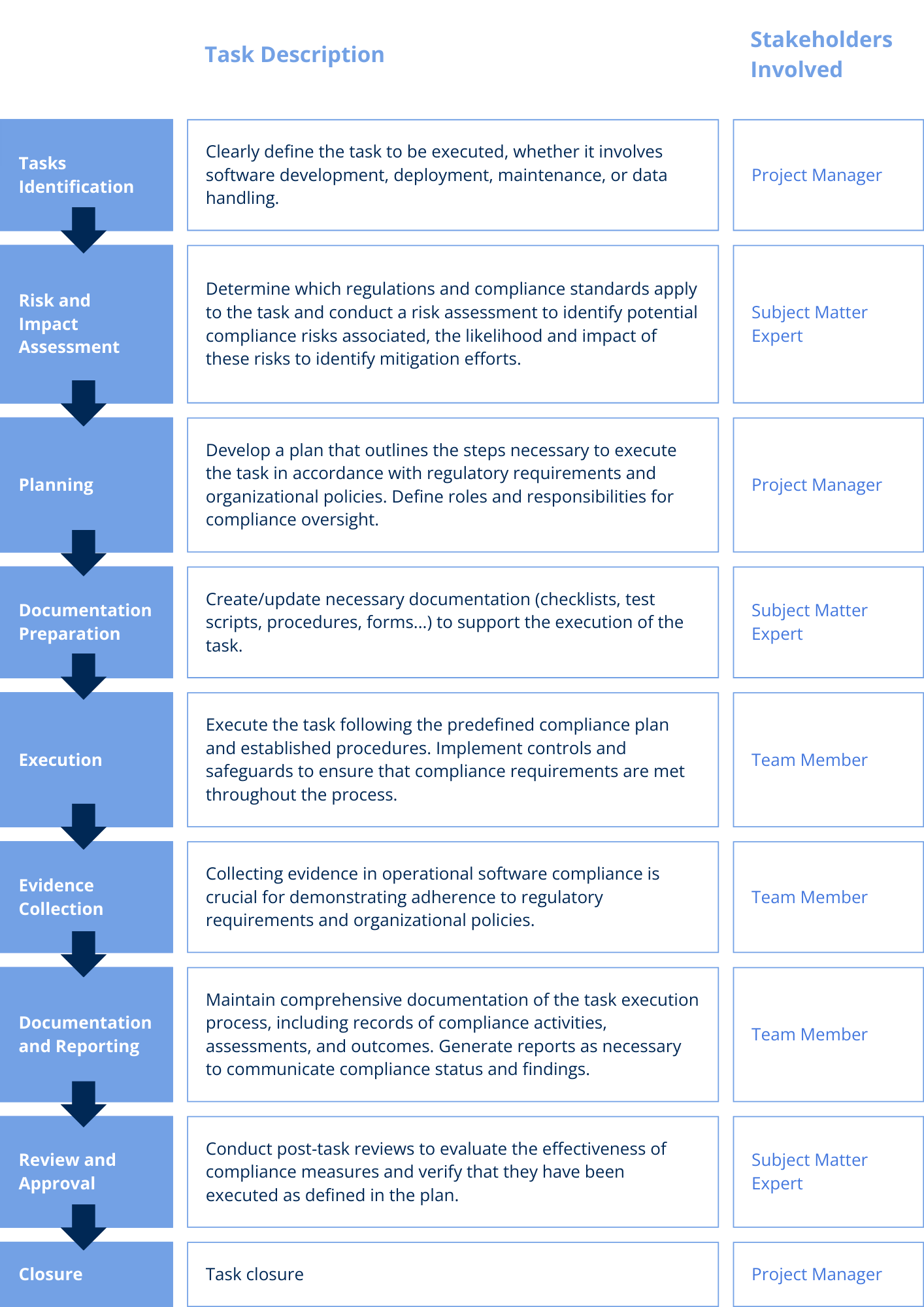
Activities in the operational phase
Tasks that may arise during the operational phase can be categorized based on their impact:
-
Recurrent Tasks: These tasks have a defined impact and are guided by the compliance framework. They can be outsourced with clear internal guidelines and monitoring.
-
Tasks Requiring Impact Assessment: More complex tasks that need individual assessment are less suitable for outsourcing and require in-depth monitoring.
Examples of each category include:
-
Recurrent Tasks: IQ/OQ/PQ, test execution, project closure, summary reports, traceability matrix, periodic review, system monitoring.
-
Tasks Requiring Impact Analysis: Change management, incident management.
Coordination between client and service provider
Effective operational software compliance often requires coordination between the system owner (client) and the service provider.
Points of contact on both sides must be well-informed of requirements and engage in regular follow-up meetings (e.g., weekly) to oversee tasks and address needs. Clear communication channels should be established to support technical queries and collaboration.
- Client Responsibilities:
- Provide guidelines or procedures, necessary training, and system access (e.g., task tracking systems like JIRA).
- Define expected time commitments, due dates, and task priorities.
- Service Provider Responsibilities:
- Ensure team members are trained, provide feedback, and address any concerns during task execution.
- Deliver feedback on task timelines, guideline content, and any additional questions or issues at the start of each task.
The better structured and documented the tasks are, the easier and more successful the outsourcing process will be. Communication channels and documentation of execution levels (e.g., remote or on-site testing) support efficiency in compliance management.
Although regular meetings are held between points of contact, there must be a communication channel that allows for technical queries to be made by the teams on both sides.
The tests and evidence required for task execution can be conducted at different levels:
-
Global systems / systems with remote access: the tester (service provider team) accesses the system remotely and performs the tests, capturing the evidence during the execution.
-
Systems without remote access and too complex to grant access: subject matter experts take evidence, and the service provider team documents the execution.
-
Systems without remote access but accessible on-site: Accessing the client's facilities to execute and collect the required evidence. Documentation of the execution can be done both on-site and remotely.
Once the task is completed and delivered, the level of review by the client should depend on the complexity of the tasks and the experience of the service provider’s team. Monitoring of KPIs will help to identify process improvement needs.

How to keep computerized systems in the operational phase?
Conclusion
The validation process does not end once the system is implemented; it is a continuous effort throughout the system's lifecycle. Key points for maintaining compliance in the operational phase include:
-
Well-Defined Processes and Responsibilities: Clear processes and defined roles ensure tasks are executed accurately, minimizing confusion and ensuring compliance.
-
Clear Priorities and Deadlines: Establishing priorities and timelines prevents delays and ensures efficient resource management.
-
Comprehensive Training: Training team members on procedures, tools, and GxP standards ensures tasks are performed accurately.
-
Utilization of Task Management Tools (e.g., JIRA, Asana): These tools help organize and track compliance tasks, fostering collaboration and accountability.
-
Continuous Follow-Up: Regular internal communication and follow-up meetings keep teams aligned and informed of any emerging compliance needs.
By following these principles, organizations can strengthen collaboration, improve efficiency, and ensure regulatory adherence in their operational software compliance practices.
Navigating operational software compliance in pharma can be complex. At QbD Group, we provide expert guidance to help you implement robust compliance frameworks, streamline processes, and ensure regulatory alignment throughout your software lifecycle.
Ready to strengthen your compliance strategy? Contact our team today to discuss how we can support your journey toward seamless, efficient operations.

About the Author
QbD Group is a global life sciences consultancy supporting companies from idea to patient. With over 700 experts, QbD delivers regulatory, quality, and compliance services across pharma, biotech, medical devices, and diagnostics.
Subscribe to the latest updates in life science
Expert perspectives delivered to your inbox — pick your interests.
No spam, ever. Unsubscribe anytime.



