
Navigate Regulatory Complexity with Confidence
Navigate complex regulatory landscapes with expert guidance across pharmaceuticals, medical devices, and IVD.
Read more订阅生命科学领域的最新动态
专家观点直达您的收件箱——选择您的兴趣。
绝无垃圾邮件。随时取消订阅。
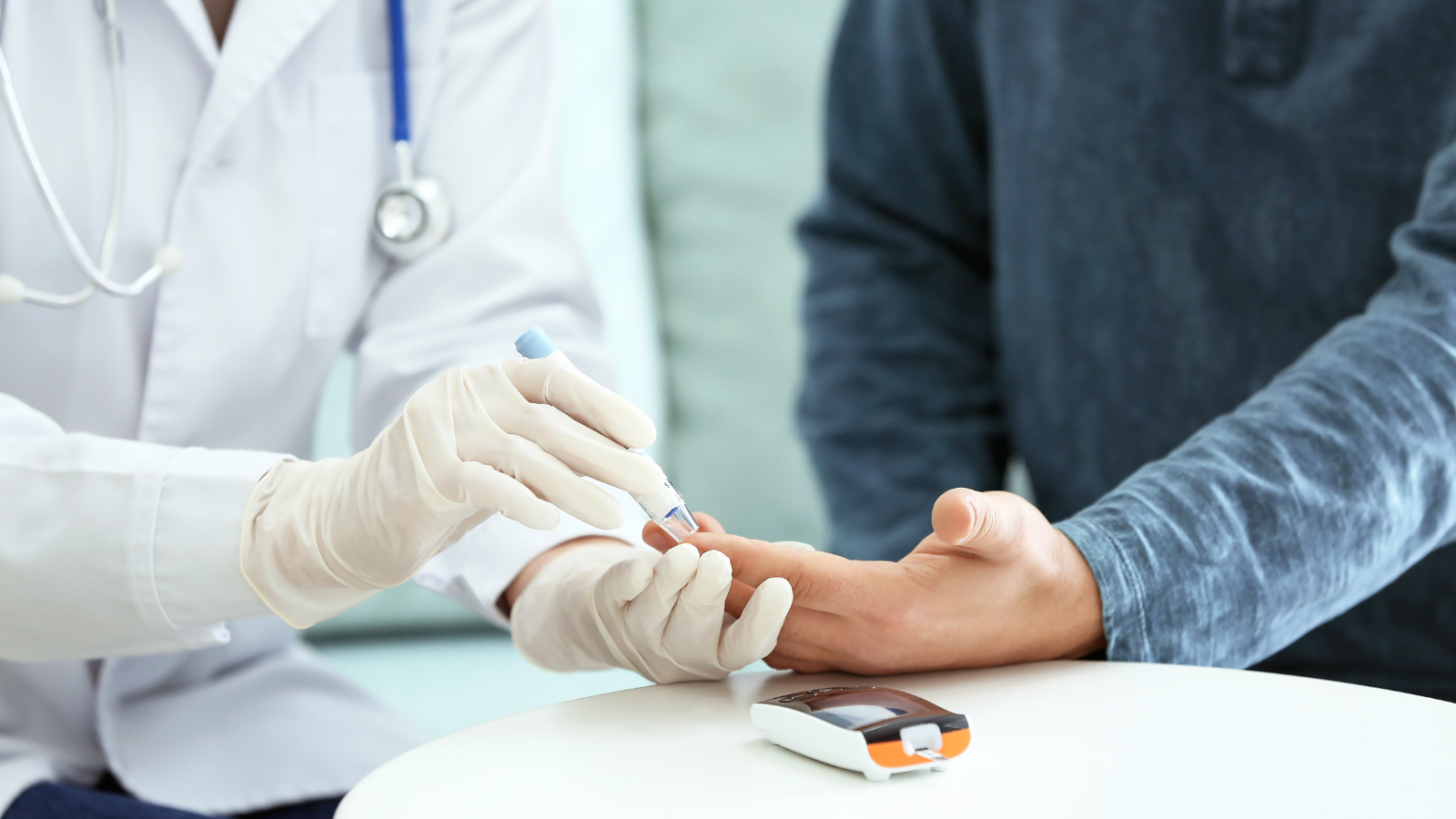
Understanding the impact of EU IVDR on clinical trials and the changes it brings. Stay informed and compliant with the latest regulations.

Navigate complex regulatory landscapes with expert guidance across pharmaceuticals, medical devices, and IVD.
Read more专家观点直达您的收件箱——选择您的兴趣。
绝无垃圾邮件。随时取消订阅。
Keep reading

Discover why governance and role clarity are critical in combined drug-diagnostic studies under the CTR and IVDR.
阅读更多
Discover why early collaboration between pharma sponsors and IVD partners is critical for successful companion diagnostic development under the IVDR.
阅读更多
Thinking Article 5(5) is the easy route under the IVDR? Discover the hidden obligations and risks of in-house devices.
阅读更多