Learn more from our article: Mastering Cleanroom Qualification: Navigating the Latest Regulatory Updates.
Equipment Qualification
Following the facility and utilities setup, the equipment qualification phase commences. This process can involve anything from simple apparatuses to intricate systems.
As illustrated in Figure 1, equipment qualification adheres to specific GxP guidelines, such as GAMP5, ISPE, and ISO standards. Customized or more complex systems undergo factory acceptance testing (FAT) and/or site acceptance testing (SAT) during the commissioning phase.
These pivotal tests, when thoroughly conducted and documented, can mitigate errors in the subsequent qualification stage. An efficient qualification not only streamlines the process but also alleviates project-end pressures.
Dive deeper into equipment qualification with our blog: Qualification of Laboratory Equipment: Key Considerations.
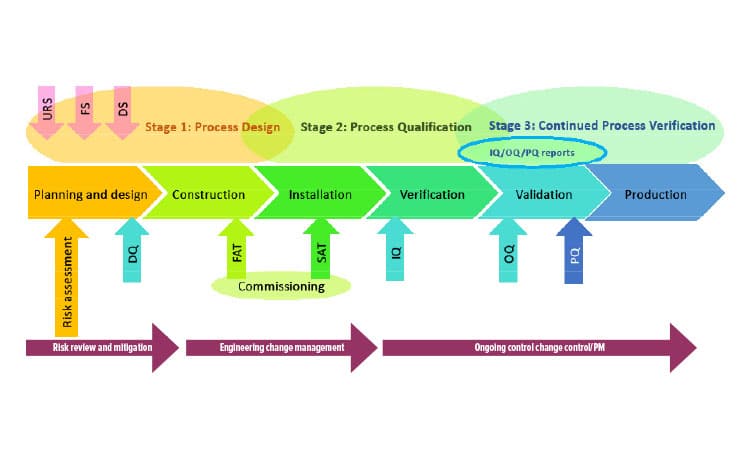
Figure 1: Lifecycle Approach to Design, Qualification, and Continued Verification ( Source: ISPE)
Thorough documentation of GMP processes and outcomes is vital for GMP compliance. The validation process essentially documents that the instruments, processes, and activities at the facility consistently produce expected results. GMP mandates process validation, cleaning and sanitation validation, computer system validation, and analytical method validation.
New GMP Facility: Pitfalls & Best Practices
In the intricate journey of GMP facility qualification, no path is without its challenges. From planning to team collaboration, this section explores the pitfalls we’ve navigated and the insights we’ve garnered, ensuring (your?) future endeavors are even more streamlined and efficient.
Plan Adaptively
From our experience in qualifying new GMP facilities, we’ve amassed a wealth of lessons. We’ve navigated pitfalls concerning planning, budgeting, and other factors. Yet, we creatively overcame these obstacles to ensure quality deliverables.
As mentioned earlier, everything stems from a predefined design. This encompasses the tasks to be done, the timeline for completion, and the personnel required to achieve the objectives. Proper planning integrates all this information. Ideally, such planning should already account for potential project delays, be they in construction, equipment delivery, or installation issues. While we cannot predict every turn of events, we must be prepared to adapt when unforeseen challenges arise.
One significant pitfall is the temptation to delay activities, particularly early in the project. While no approach is flawless, it’s vital to establish clear agreements from the outset about how to handle such shifts. Postponing tasks can risk overshooting the budget and missing deadlines.
It’s advisable to only delay activities when the installation hasn’t been scheduled or when it doesn’t affect other operations. If not, consider rearranging activities in your plan. When one task gets postponed due to unforeseen delays, another can be expedited.
Set Realistic Timelines & Budgeting
To ensure a smooth process without unnecessary adjustments, it’s essential to establish a realistic timeline from the onset. A deep understanding of the activities involved is crucial for accurate timeline estimation.
This necessitates a multidisciplinary team equipped with insights into construction, the impact of installations, qualification, validation, and a clear understanding of the resources required for each activity, both in terms of time and budget.
Beyond just the resources needed for the main tasks, supporting departments such as production, maintenance, calibration, and especially QA play vital roles in providing input, conducting reviews, and approving expected deliverables.
As previously emphasized regarding planning, building in flexibility is essential when setting up a new GMP-compliant facility. This allows for adjustments in response to unforeseen delays. Avoid setting rigid milestones, where feasible, to prevent creating a high-pressure, stressful working environment.
Set and Celebrate Achievable (!) Milestones
We’ve addressed planning and timelines. To mark progress and foster a sense of achievement, it’s vital to set realistic milestones throughout the project. These can signal the onset of a new project phase or the qualification and release of the first cleanroom for use. Achieving these milestones boosts team morale and drives motivation.
However, it’s crucial that these milestones are attainable. Setting unachievable targets can lead to feelings of defeat, dampening the team’s overall spirit. Beginning a project with the mindset of “we know it’s nearly impossible, but you must achieve it” is not the ideal approach, especially for high-pressure projects.
Communicate and Cooperate Effectively
A project isn’t a one-man show. Achieving a qualified GMP facility requires the collective expertise of many, spanning technical know-how, GMP qualification, quality assurance, procurement, finance, and other resources. Effective communication from the outset ensures all departments align their goals, intentions, and understanding of each other’s expertise.
In projects with strict timelines, appointing a SPOC(single point of contact) for each team is essential to facilitate seamless two-way communication. Such a step fosters appreciation and respect.
When concerns arise, it’s crucial to address them rather than dismiss them; unattended issues might escalate as the project progresses. Prevention is always preferable to resolution.
Need Help with Your New GMP Facility Qualification?
Setting up a new GMP facility involves a multitude of tasks that can seem overwhelming to ensure GMP compliance for commercial operations. Our seasoned experts are poised to assist, from advisory roles to managing and executing all qualification processes.
Are you planning on a new GMP facility? New to qualification? Or do you need advice? Our experts will be happy to help you with the best validation program in accordance with your available resources.

关于作者
订阅生命科学领域的最新动态
专家观点直达您的收件箱——选择您的兴趣。
绝无垃圾邮件。随时取消订阅。



