Digital therapeutics (DTx) represent an exciting frontier in healthcare, leveraging clinically validated digital technologies to enhance health and well-being. DTx offers a broad spectrum that includes behavioral interventions, systems incorporating wearable devices, artificial intelligence, mobile tech innovations, and big data analytics.
This introductory blog post is the first in a series in which we will shed light on the rise of DTx, its regulatory environment, the perspective of reimbursement payers, and the overarching vision of the World Health Organization.
Digital Health Tools at a Glance
Ranging from tools for medication adherence and health goal setting to self-monitoring tools for conditions like hypertension and diabetes, DTx encompasses a myriad of apps, digital systems, and web portals.
These tools have the profound ability to bridge care gaps, especially for underserved or remote populations, and potentially curtail overall healthcare delivery costs in our financially strapped health ecosystem.
Safety is paramount when integrating digital solutions into healthcare. Concerns include the risk of misinformation, computational errors, misdiagnoses, or unsuitable patient solutions.
To address these concerns, DTx providers must prove quality performance, clinical compliance, and robust data protection. Health authorities translate these necessities into intricate regional and national regulations.
In upcoming blog posts, we will explore specific regulations in regions like the EU and countries including Germany, Belgium, France, the UK, and the US, highlighting their impact on DTx providers.
Reimbursement Payers’ Perspective
Beyond safety, showcasing economic and quality-of-life benefits is pivotal for reimbursement considerations. For instance, in cardiology, patients benefit from consistent remote monitoring outside clinical settings. However, reimbursement models vary nationally and are influenced by the specific disease focus.
The Expanding Realm of DTx
Currently, DTx solutions primarily operate as independent software. Yet, the idea of integrating DTx with traditional treatments, such as medications, has sparked interest across both budding digital health startups and major tech giants, even those outside conventional healthcare sectors.
In the table beneath, you’ll find all types of examples of digital therapeutics within the bigger picture.

Figure 1 – CRA referencing Deloitte’s 2021 Report on Digital Therapeutics
WHO’s Vision for Digital Health
The World Health Organization ( WHO) champions the advancement of digital health technologies. Their ‘Global Strategy on Digital Health 2020-2025’ serves as a beacon, guiding nations towards broadening digital health services.
The strategy’s ambition is clear: enhance global access to personalized digital health solutions that can effectively prevent, detect, and manage health threats, including epidemics and pandemics.
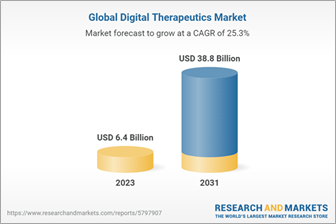
Encouragingly, the strategy propels health authorities to define market access rules and set reimbursement standards. With the WHO’s endorsement, the digital health market’s future looks promising, with projections indicating a 25.3% growth from 2023 to 2031.
Figure 2 – Frontiers in Medtech
Conclusion
Digital therapeutics, blending seamlessly into our digital lives, hold a transformative promise for healthcare. Not only have they been integrated into society’s fabric, like smartphones and tablets, but institutions like WHO also laud their potential in combating global health challenges. In upcoming blog posts, we’ll navigate the regulatory intricacies these innovations face.

with ease.

Navigate Regulatory Complexity with Confidence
Navigate complex regulatory landscapes with expert guidance across pharmaceuticals, medical devices, and IVD.
Read more订阅生命科学领域的最新动态
专家观点直达您的收件箱——选择您的兴趣。
绝无垃圾邮件。随时取消订阅。