
How to avoid non-compliance in the pharmaceutical industry
Build a successful compliance program in pharma by strengthening internal controls and procedures to ensure robust regulatory adherence.
阅读更多.png?width=1080&height=1080&name=Salma%20Michor%20(2).png)
PhD in Bio Engineering, MSc Food & Biotechnology, MSc King's College London, MBA Open University, RAC (EU & Global), Chartered Manager, RAPS European Board Member · Division Head Regulatory Affairs
Salma Michor is currently Division Head Regulatory Affairs at the QbD Group. She served clients such as Johnson & Johnson, Novartis, Pfizer and CSL Behring. Previously, Michor worked for Chiesi-Torrex, Wyeth Whitehall Export Croma Pharma GmbH. She teaches regulatory affairs and clinical strategies at the University of Krems, Austria, and is an independent expert to the European Commission.
2 articles

Build a successful compliance program in pharma by strengthening internal controls and procedures to ensure robust regulatory adherence.
阅读更多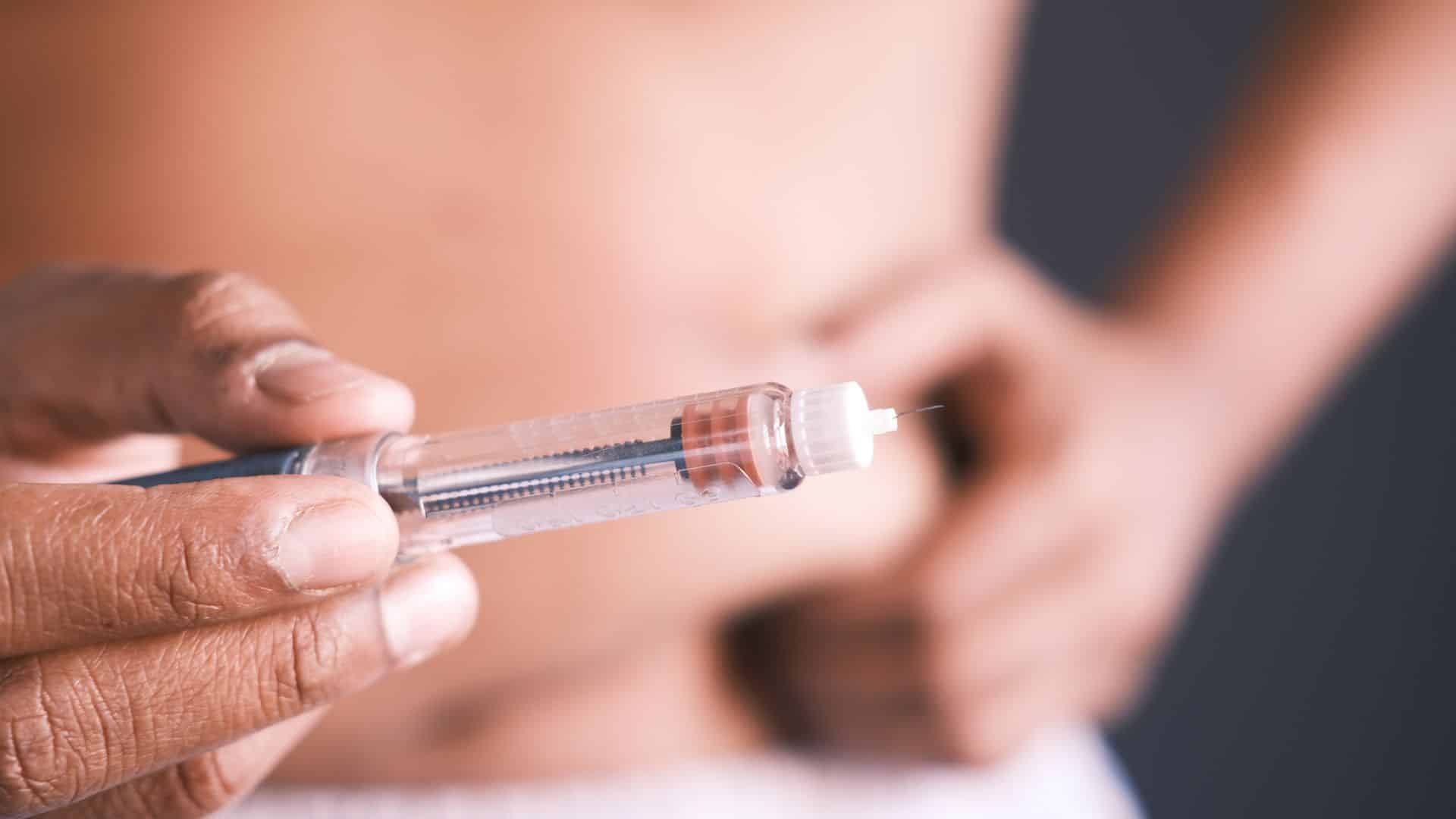
Discover how to navigate compliance with Article 117 of MDR for drug-device combinations. Ensure correct implementation with essential guidance.
阅读更多