Pharmaceutical, clinical, MD, and IVD companies pursuing robustness in their software validation activities can use either the V-model or the Agile model. Both are Computer System Validation (CSV) methods that can be applied according to GAMP 5.
The Good Automated Manufacturing Practice (GAMP) framework is a set of guidelines that provide a structured approach to validation in regulated industries, including pharmaceuticals, biotechnology, and medical devices. GAMP 5 is the latest version of this framework, released in 2008 and updated in 2022.
This blog post is about the Agile model and is a follow-up to the V-model blog post we shared earlier.
Enjoy reading!
A complete guide to Computer System Validation
This +100 page guide aims to bring context and define the necessary and appropriate strategies for the validation of computerized systems for Pharmaceutical Industries, Biologics, Biotechnology, Blood Products, Medicinal Products, and Medical Devices, used in activities related to compliance with Good Practices (GxP). Download it now for free:
Why validate your computerized systems?
Using software for your life science activities requires a high level of integrity and security in the data processing. The computerized system must be designed to operate in a consistent and reproducible manner, in accordance with applicable GxP regulations, to ensure the expected final product quality. Validating this system provides documented evidence that it does exactly what it is intended to do. In order to achieve this, you can use either the V-model or the Agile model.
The Agile model in CSV
The Agile model is based on a continuous exchange between the client and the validation team. The stages of the project rely on constant feedback from the customer to make progress.
Agile development methods have become increasingly popular in recent years because they offer a flexible and iterative approach to software development that can help teams quickly deliver high-quality products.
However, it can be challenging to validate agile development processes in regulated industries, where strict quality standards must be met to ensure patient safety.
The V-model in CSV
In a V-model, the ‘V’ can stand for validation and verification, but it also refers to the shape of its common graphical representation.
It is a variant of the Waterfall model of software development, which describes a sequential procedure in which each step must be completed before the next can be started. Since cycles are not possible here, the V shape appears.
Validation according to the Agile model
Agile development is an iterative approach to software development that emphasizes collaboration, flexibility, and rapid delivery. Unlike traditional software development methods following the V-model or Waterfall approach – which involve a sequential process of planning, designing, coding, testing, and deployment – the Agile model focuses on delivering working software in small steps, with frequent feedback from users and stakeholders.
While this approach can be very effective for developing software quickly, it can be challenging to validate in regulated industries.
Validation is the process of demonstrating that a product or process meets predetermined requirements and specifications and is suitable for its intended use. In regulated industries, validation is required to ensure patient safety, product quality, and compliance with regulatory standards.
To validate agile development processes, organizations must demonstrate that they have a robust quality management system in place that can manage the risks associated with agile development. This includes implementing appropriate controls, procedures, and documentation to ensure software meets regulatory requirements.
GAMP 5 framework for agile development
The GAMP 5 framework provides a structured approach to validation that can be applied to agile development processes. The framework is based on a risk-based approach, identifying potential risks and implementing appropriate controls to mitigate those risks.
The GAMP 5 framework consists of 4 categories:
- Category 1 – Infrastructure software
- Category 3 – Non-configured products
- Category 4 – Configured products
- Category 5 – Customized applications
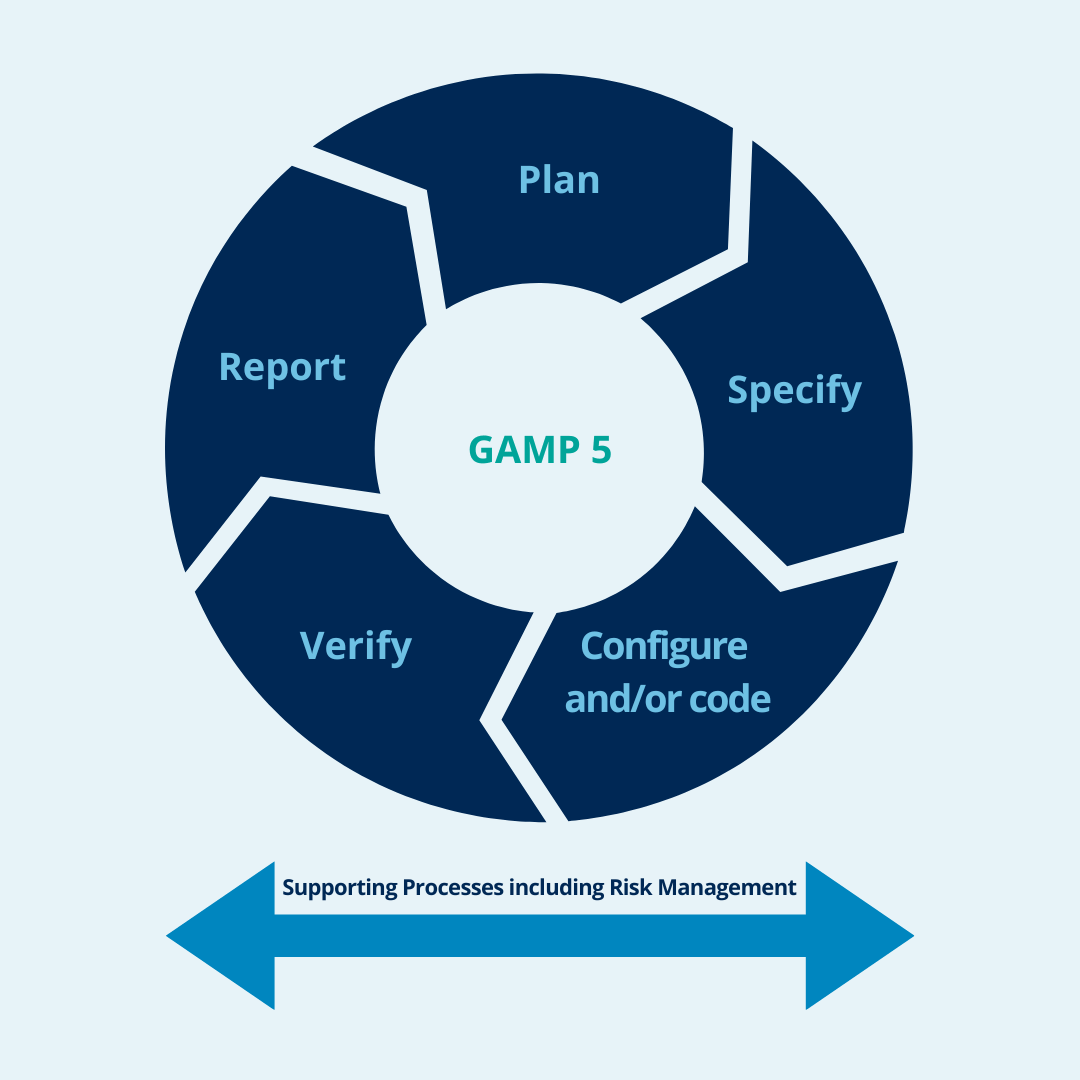
To validate agile development processes according to GAMP 5, organizations must follow the framework’s five-phase approach:
Planning
Determine the scope and strategy of the validation effort and develop a validation plan that includes identification of critical processes and requirements.
Specify
The delivered description of system functionality is provided by the list of completed agile artifacts, such as epics and user stories, taken directly from agile software development tools.
Configure and/or code
In this step, the supplier relies on its expertise to implement the customer’s requirements. All functionalities documented in the previous steps are implemented.
Verify
Verify that the software meets user requirements and design specifications through testing and other validation activities, such as peer reviews and code inspections.
Report
Develop a design specification that describes how the software will be designed, including the software architecture, data model and interfaces.
In agile development, these 5 phases are typically conducted in iterative cycles, with each cycle resulting in a working software increment. At the end of each cycle, the software increment is validated to ensure that it meets the user requirements and design specifications.
Special considerations when using the Agile model
Integral part of the development process
In an agile environment, validation must be an integral part of the development process, with validation activities performed in each sprint. This approach allows validation to be built into the development cycle, rather than being an afterthought or bottleneck.
One way to achieve this is by using automated testing tools that can validate functionality quickly and reliably. Automated testing tools can be integrated into the development cycle, allowing testing to be performed automatically after each code change.
Documentation
Another important consideration when validating computerized systems in an agile environment is documentation. Regulatory requirements typically require that computerized systems be documented thoroughly, including requirements, design, testing, and maintenance.
In an agile environment, documentation should be incorporated into the development cycle, with documentation updated in each sprint. Documentation should be clear, concise, and easy to understand, allowing developers, testers, and stakeholders to understand the system’s functionality and validate that it meets the specified requirements.
Communication
Communication is also essential when validating computerized systems in an agile environment. Agile development emphasizes collaboration and communication, and this applies to validation as well.
Developers, testers, and stakeholders should communicate frequently, with regular feedback loops throughout the development cycle. This approach allows issues to be identified and addressed quickly, reducing the risk of delays or failures.
Validation strategy
Finally, it’s important to establish a validation strategy and plan before starting development. The validation strategy should outline the validation approach, including the scope, objectives, and acceptance criteria.
The validation plan should provide a roadmap for validation activities, including testing, documentation, and communication. These documents should be updated throughout the development cycle, reflecting changes in the system’s functionality or regulatory requirements.
Conclusion: use GAMP 5 and the Agile model to your advantage
Validating agile development processes in regulated industries requires a risk-based approach that considers the unique challenges and complexities of agile development.
The GAMP 5 framework provides a structured approach to validation that can be applied to agile development processes, enabling organizations to develop high-quality software products that meet regulatory requirements and ensure patient safety. By following the five-phase approach outlined in GAMP 5, organizations can validate their agile development processes – following the Agile model – whilst being compliant.
In conclusion, validating computerized systems following the Agile model requires a proactive and collaborative approach. By incorporating validation into each sprint, using automated testing tools, communicating frequently, and documenting thoroughly, developers, testers, and stakeholders can work together to ensure that computerized systems meet regulatory requirements and are fit for their intended use.
With these best practices in place, organizations can successfully implement agile development methodologies while maintaining compliance with regulatory requirements.
Need help and assistance? QbD is here for you! Don’t hesitate to contact us to come up with a pragmatic and customized approach for your business.