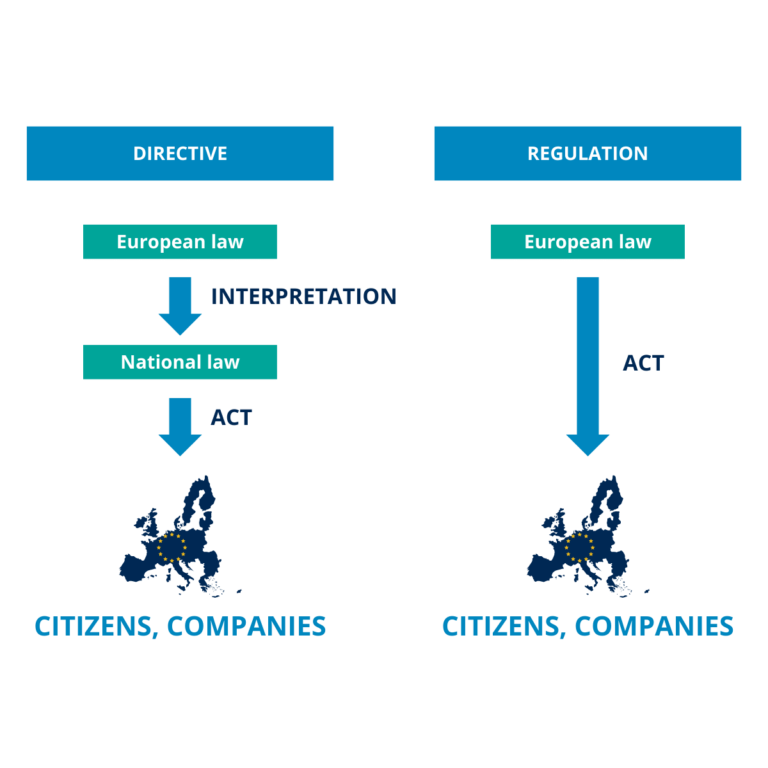
On 26 April 2023, the European Commission adopted a proposal for a new Directive and a new Regulation that revise and replace:
- the existing general pharmaceutical legislation (Regulation 726/2004) and Directive 2001/83/EC),
- the legislation on medicines for children (Regulation 1901/2006),
- and for rare diseases (Regulation 141/2000/EC)
This general reform of pharmaceutical legislation would be the largest reform in over 20 years – to make it more agile, flexible, and adapted to the needs of citizens and businesses across the EU. The revision will make medicines more available, accessible, and affordable. It will support innovation and boost the competitiveness and attractiveness of the EU pharmaceutical industry while promoting higher environmental standards.
Via this reform, the European Commission is therefore proposing to modernize the pharmaceutical sector with a patient-centered approach, that also fully supports an innovative and competitive industry. Its approach will preserve the EU’s high standards for the authorization of safe, effective, and quality medicines. It aims to achieve the following main objectives:
- Drastically reduce the administrative burden by speeding up procedures significantly, reducing authorization times for medicines, so they reach patients faster;
- Create a Single Market for medicines ensuring that all patients across the EU have timely and equitable access to safe, effective, and affordable medicines;
- Continue to offer an attractive and innovation-friendly framework for the research, development, and production of medicines in Europe;
- Enhance availability and ensure medicines can always be supplied to patients, regardless of where they live in the EU;
- Address antimicrobial resistance (AMR) and the presence of pharmaceuticals in the environment through a One Health approach;
- Make medicines more environmentally sustainable.
These adopted acts are open for feedback for a period of 8 week(s). All feedback received will be summarised by the European Commission and presented to the European Parliament and Council with the aim of feeding into the legislative debate. Via this route, the regulation and directive will have to go through the EU’s legislative process before being approved, it’s unlikely this will be done before the European Parliament elections in 2024.
QbD Group will continue to monitor the progress of this reform and continue to update our customers on the final text which will be implemented after review and adjustments by the EU Parlement in Council. Especially towards the regulatory framework and the aim of reducing the administrative burden for medicines to ensure our customers remain updated on any different expectations in the future in the way of working for the regulatory affairs departments of the customers of QbD Group.