The pharmaceutical industry has seen its fair share of revolutions. From early mechanisation to computerised systems, each wave of innovation has reshaped the way medicines are developed and delivered. Today, we’re entering a new era: Pharma 4.0 — a data-driven, highly connected model that builds on the foundation of Industry 4.0 to redefine how the industry operates.
But what exactly is Pharma 4.0? How does it differ from previous models? And what does it mean for companies navigating ever-increasing demands for efficiency, compliance, and patient-centricity?
Let’s explore the paradigm shift that is reshaping pharma — and why preparing for it now can give your business a critical edge.
From Pharma 3.0 to 4.0: a shift in perspective
Since the second half of the 20th century — and especially during the early 2000s — pharmaceutical production methods have been influenced by the third industrial revolution. This period, often referred to as Pharma 3.0, was marked by increasing digitisation and automation across the production cycle. Companies began using digital tools to control and optimise industrial processes, improving product quality and operational consistency.
However, while Pharma 3.0 introduced critical improvements, it also left some gaps — particularly in information management. Although data was being generated, it was rarely analysed or used in real time. Many systems remained disconnected, limiting the full potential of digitisation.
This led to a process-centric approach. Most decisions were tied to workflow efficiency rather than to the strategic use of data. Automation helped optimise operations, but decision-making remained bounded by predefined processes.
Enter Pharma 4.0: a model that moves from process-driven thinking to data-driven intelligence. In this new paradigm, data is not just a by-product — it becomes the foundation for operational, strategic, and business decisions. Technologies like AI and Big Data make it possible to collect, analyse, and act on vast volumes of information in real time, reshaping the way pharmaceutical companies innovate and operate.
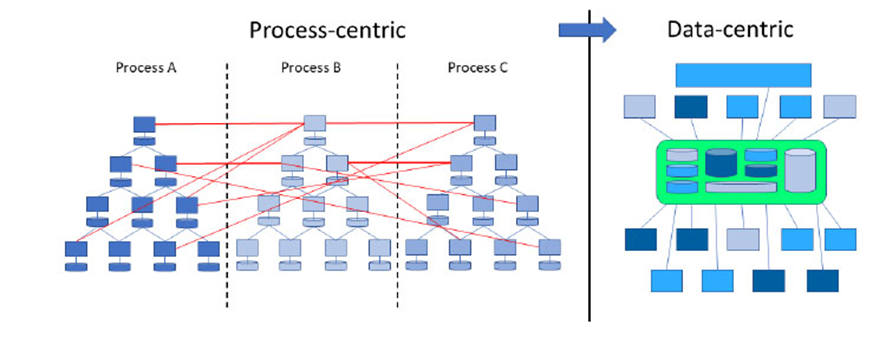
Image obtained from ispe.org
What Is Pharma 4.0?
Pharma 4.0 is the pharmaceutical industry's response to Industry 4.0 — the broader industrial transformation towards smart, interconnected, and autonomous systems. It involves the integration of:
- Advanced automation
- Artificial Intelligence and Machine Learning
- Internet of Things (IoT)
- Cloud computing and cyber-physical systems
- Big Data analytics
Together, these technologies form a fully digital ecosystem, where equipment, systems, and processes are interconnected, validated, and capable of learning and adapting continuously.
| Pharma 3.0 | Pharma 4.0 | |
| Technologies | Basic digitisation, limited automation | AI, IoT, Big Data, advanced automation, real-time connectivity |
| Approach | Process-centric | Data-centric |
| Focus | Process efficiency and product quality | Predictive analytics, optimisation, personalisation |
| Decision-making | Based on production workflows | Based on real-time, data-driven insights |
| Data usage | Collected but underutilised | Analysed and actioned continuously |
Pharma 4.0 doesn’t just improve how we do things — it changes what becomes possible.
The Benefits of Pharma 4.0
Although the model is still being implemented across the industry, the benefits of Pharma 4.0 are already tangible:
1. Smarter, more connected manufacturing
IoT devices and automation enable real-time monitoring and adjustment throughout the production process. Smart manufacturing systems can identify deviations, make autonomous corrections, and ensure consistent product quality.
2. Continuous optimisation
Advanced analytics and AI help detect inefficiencies and predict maintenance needs or process improvements. This allows companies to fine-tune operations continuously, reducing downtime and waste.
3. Faster, more effective drug development
Digital tools like in silico trials, molecular modelling, and AI-assisted discovery speed up R&D. Entire libraries of drug candidates can be screened digitally, significantly reducing development time and cost.
4. Increased personalisation
The use of Big Data enables personalised medicine based on genetic, demographic, or behavioural data. Treatments can be tailored to individual patients, improving efficacy and outcomes.
Challenges and risks
While the potential is vast, the shift to Pharma 4.0 is not without hurdles:
- Cybersecurity vulnerabilities
Greater connectivity means greater exposure. Secure data infrastructure and access controls are crucial.
- High implementation costs
The transformation involves not only technologies, but also significant investment in change management, recruitment, and training.
- Technological dependency
Heavy reliance on digital systems increases the impact of technical failures. Robust contingency planning is essential.
These risks are real, but with the right preparation, they can be mitigated. And the benefits far outweigh the challenges for companies willing to invest in the transition.
Pharma 4.0 creates new growth opportunities
Pharma 4.0 doesn’t just improve existing operations — it opens doors for innovation:
- Accelerated R&D
Advanced modelling and high-throughput digital screening enhance the discovery of new molecules.
- More flexible, efficient production
Smart manufacturing allows batch adjustments in real-time and better resource utilisation.
- New tech markets
The need for robust software, cloud infrastructure, and compliant automation tools creates opportunities for tech providers and service companies.
- Regulatory advantage
Companies that implement validated, traceable digital systems are better positioned to meet evolving compliance requirements.
Conclusion: planning for the future of pharma
Pharma 4.0 marks a turning point in how pharmaceutical companies operate. By embracing digitalisation and data-centric models, businesses can improve quality, reduce waste, and ultimately deliver better outcomes for patients.
But the transition isn’t just about buying new tools. It’s about building a smart, scalable system where technology, people, and processes work in harmony.
At QbD Group, we support pharmaceutical companies in every stage of their Pharma 4.0 journey — from system validation to digital transformation strategies.
Need expert support to future-proof your operations?
Let’s talk about how we can help.

Subscribe to the latest updates in life science
Expert perspectives delivered to your inbox — pick your interests.
No spam, ever. Unsubscribe anytime.



