In vitro diagnostics (IVD)
From idea to patient — your IVD industry expert
The in vitro diagnostics (IVD) industry is evolving rapidly, driven by technological innovations, complex regulatory landscapes, and the increasing need for global market access. At QbD Group, we help IVD manufacturers overcome these challenges with tailored solutions — from regulatory affairs and quality assurance to clinical evidence and technical documentation.
Comprehensive IVD services
We provide end-to-end support across the entire IVD lifecycle including CRO services, IVDR transition support, authorized representation, and more.
Regulatory expertise
Deep knowledge of IVDR, IVDD, and global IVD regulations to navigate complex compliance requirements.
Clinical evidence generation
Clinical performance studies compliant with IVDR, Common Specifications, WHO Prequalification, and ISO 20916.
MedTech startup support
Tailored RA, QA, clinical, and software expertise to bring your IVD start-up from idea to patient.
Tailored solutions
Our solutions for In vitro diagnostics (IVD)
Explore purpose-built solutions designed to address the specific regulatory, quality, and compliance challenges in your industry.
Cross-industry solutions
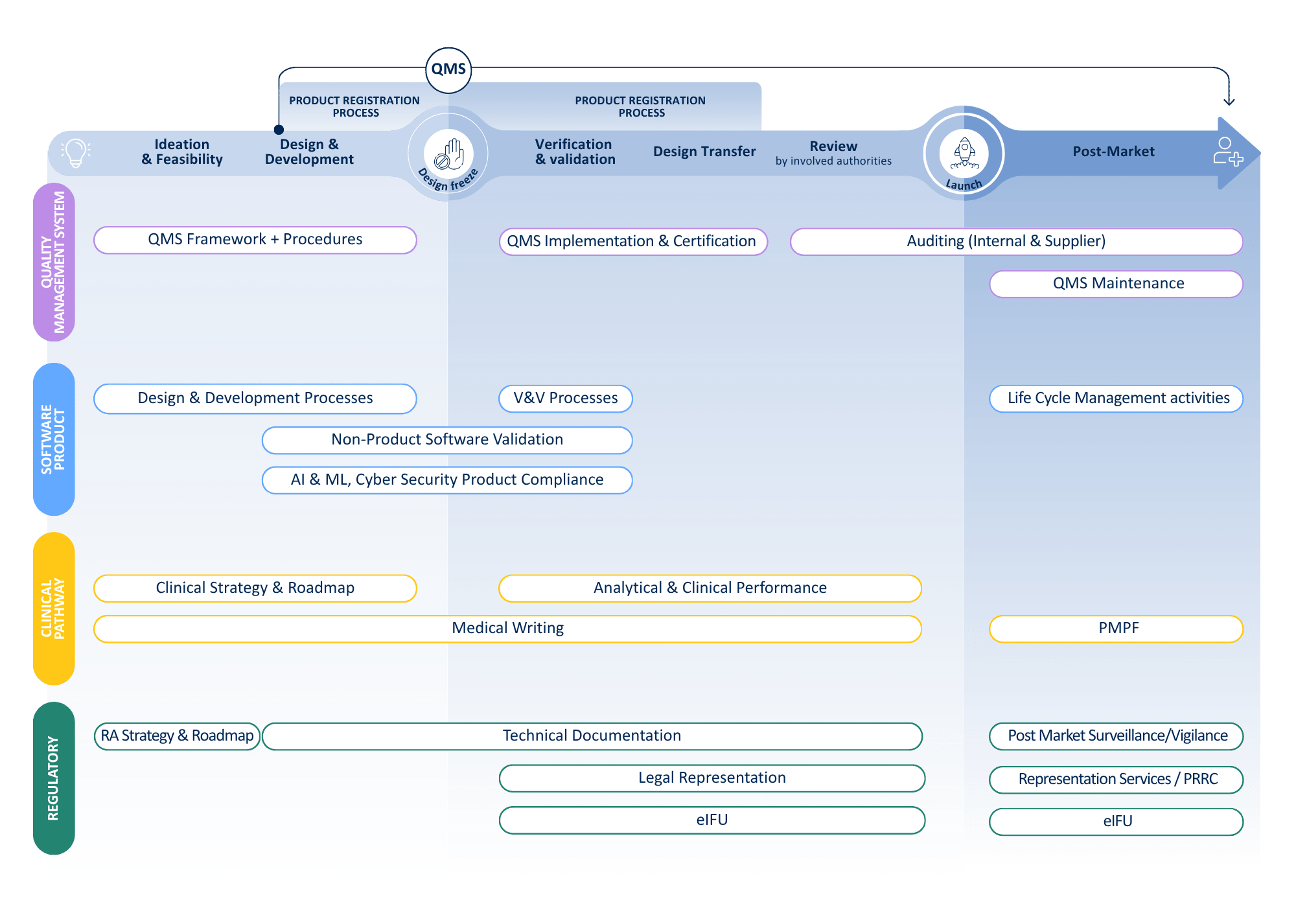
Lifecycle
Product lifecycle
We cover the full IVD life cycle — from regulatory expertise to quality assurance and clinical evidence.
Industry context
Industry challenges
These are the realities shaping your industry — and exactly where our specialists make a difference.
IVDR transition
Adapting to new regulatory requirements, securing notified body approval, and ensuring compliance with stringent clinical evidence standards.
Market access
Managing product launch timelines while keeping up with evolving regulations.
Companion Diagnostics (CDx)
Addressing the growing need for precision medicine tools.
Balancing innovation & compliance
Navigating advancements in AI-driven diagnostics, Medical Device Software (MDSW), and digital health within regulatory frameworks.
Data integration & security
Ensuring compliance with data integrity requirements while leveraging new technologies.
Services
Featured12

Why QbD Group
Your trusted partner
With decades of expertise in IVD, we deliver solutions that drive success. Our team has extensive IVDR certification experience and strong relationships with notified bodies. Over 100+ IVDR Technical Documentations completed since 2018.
Unmatched Regulatory Expertise
a team with extensive IVDR certification experience and strong relationships with notified bodies.
Proven Track Record
over 100+ IVDR Technical Documentations completed since 2018.
Customized, High-Quality Solutions
strategies tailored to reduce submission timelines by 25%-50%.
Full-Service CRO for IVD
24 years of experience, 280+ clinical performance studies across 45+ analytes.
FAQ
Frequently asked questions
Latest insights
Related content

Get in touch with IVD experts
Work with QbD Group to ensure your in vitro diagnostic products meet regulatory requirements and reach the market efficiently.








