Combination Products
Navigating EU and US regulatory pathways for combination products
Navigating regulatory pathways can often feel like a maze, leading to uncertainty and potential pitfalls in the submission process. This is even more so when it comes to combination products.
Nothing is more disheartening than investing significant time and resources into developing your combination product, only to face rejection from regulatory authorities due to unclear expectations or insufficient data.
At QbD Group, we offer comprehensive solutions to navigate the complex process of registering these innovative products in both the European Union (EU) and the United States (US).
Regulatory framework
In Europe, combination products are regulated based on their Primary Mode of Action (PMOA) — whether the product acts primarily as a medicinal product or a medical device. This determines which regulatory pathway applies.
Key challenges we address
Dual regulatory frameworks
Combination products must satisfy both pharmaceutical and device regulatory requirements — under MDR in Europe and FDA oversight in the US — often with different authorities and procedures.
Primary Mode of Action uncertainty
Determining whether your product is device-led or drug-led defines the entire regulatory pathway, and misclassification can result in costly delays or rejection.
Cross-functional coordination
Aligning drug, device, and packaging teams across different quality systems, testing requirements, and development timelines.
Complex submission requirements
Integrated regulatory dossiers must satisfy multiple authorities — from Notified Bodies and EMA opinions in the EU to FDA center designations and marketing applications in the US.
Device-led Combination Products
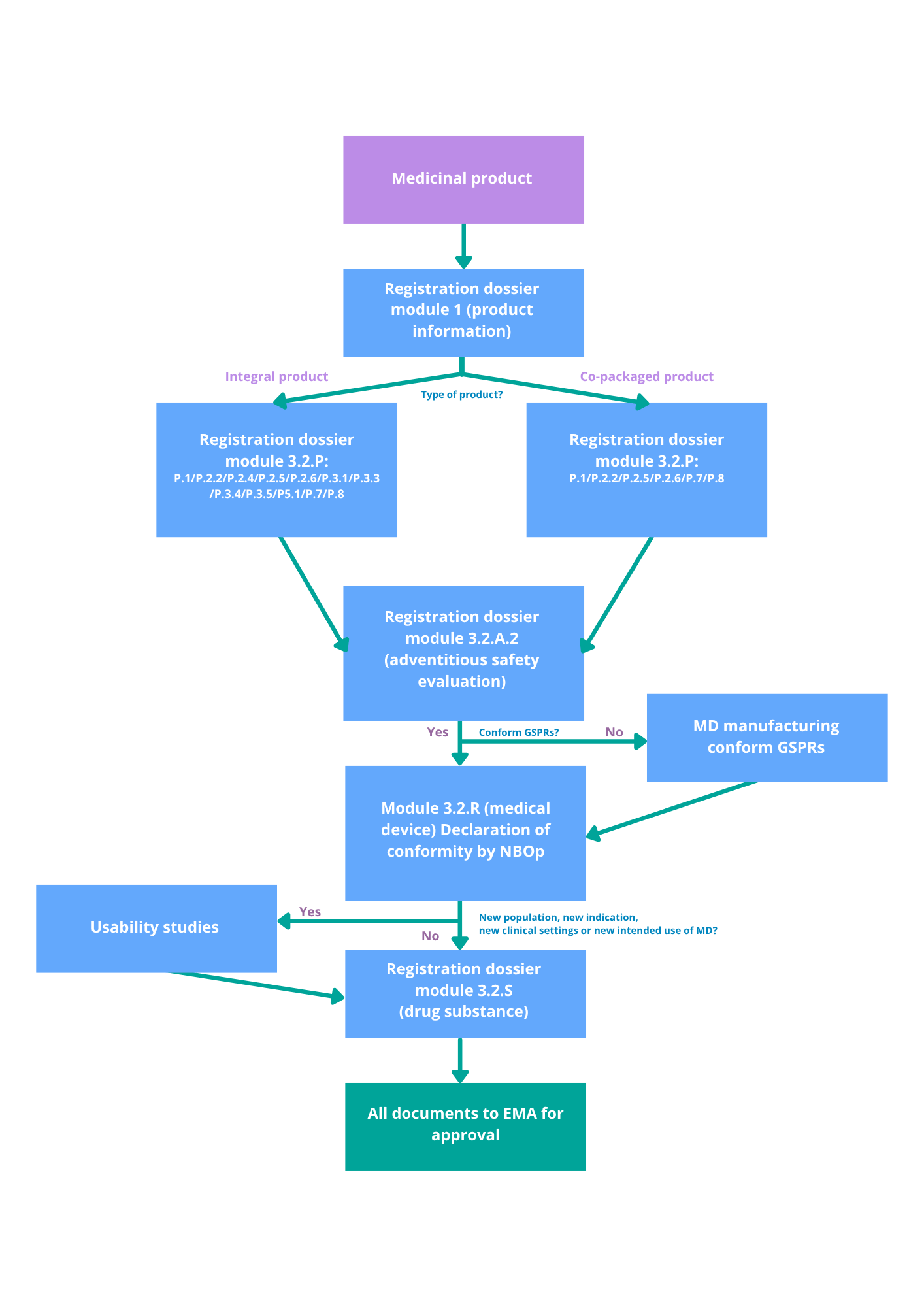
If your product is classified as a medical device with an ancillary medicinal substance, it must comply with EU MDR 2017/745. The Notified Body must consult the EMA for a scientific opinion on the excipient if it is derived from human blood or plasma, has been previously assessed by the EMA, or falls under the centralized procedure. In other cases, the Notified Body may consult either the National Competent Authority or the EMA.
Examples
Drug-eluting stent → Device pathway (MDR + EMA opinion)
Coated catheter → Device pathway (MDR)
Medicinal Product-led Combination Products
If your product is primarily a medicinal product, the combination is regulated under the Medicinal Product Directive (2001/83/EC). The type of product — and how the medical device is integrated — influences the required documentation and regulatory steps.
Examples
Prefilled syringe → Drug pathway (MPD)
Contraceptive patch → Drug pathway (MPD)
Our structured approach
Identify the lead FDA center
Determine the Primary Mode of Action (drug, device, or biologic). If unclear, submit a Request for Designation (RFD) to the FDA.
Determine the investigational application
Depending on the PMOA, apply for an IND (Investigational New Drug Application) when the primary mode is drug or biologic, or an IDE (Investigational Device Exemption) when the primary mode is device. Consider the combination product as a whole.
Select the marketing application
Drug-led: NDA or ANDA. Biologic-led: BLA. Device-led: PMA, De Novo, or 510(k). The marketing application depends on the constituent part with the PMOA.
Post-market lifecycle management
Maintain ongoing compliance across both regulatory frameworks, including vigilance, PMS, and lifecycle management for your combination product.
Our integrated offerings
Our integrated offerings support organizations across their full operational lifecycle, from compliance activities to sustained control, improvement, and digital enablement.
What you gain
Tailored regulatory strategy
We help you develop a custom regulatory roadmap to ensure your product meets all compliance and market entry requirements.
Expert guidance on procedures
Our team guides you in selecting the most suitable regulatory procedure for your product, saving time and minimizing hurdles.
End-to-end lifecycle management
From concept to market launch, we offer holistic support for every stage of your combination product's journey.
In-depth regulatory knowledge
Leverage our extensive expertise to navigate stringent requirements confidently and accelerate your product's market readiness.
Why QbD Group
Your trusted partner
Cross-industry expertise
our regulatory specialists cover Pharma, Biotech, Medical Devices, IVD, and Combination Products under one roof.
Strategic regulatory planning
we align your regulatory pathway with commercial objectives to accelerate time-to-market.
Proven track record
with thousands of successful submissions and dossier management projects across Europe, we deliver results you can count on.
Scalable support
from a single regulatory question to full outsourced regulatory operations, we adapt to your needs.
FAQ
Frequently asked questions
Related insights
View all
Have questions about the regulatory pathways for your combination product?
We understand the complexities of navigating EU and US regulations. Whether you're seeking clarity on the Primary Mode of Action (PMOA), determining the right regulatory pathway, or managing lifecycle requirements, our experts are here to help.