Quick response
Address resourcing needs as they arise, avoiding project delays

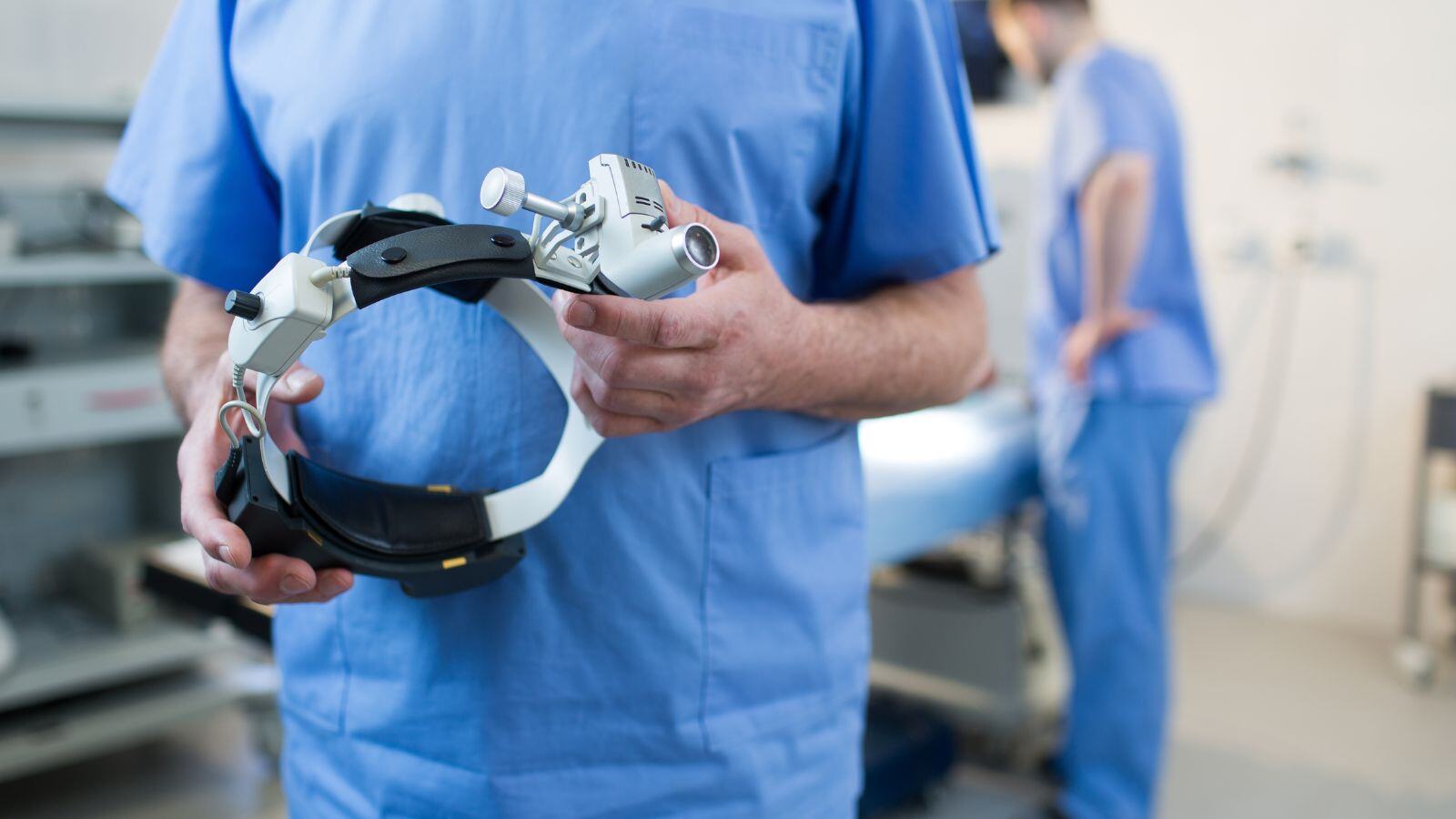
Running a clinical study comes with unique challenges, and finding the right talent shouldn’t be one of them.
Are you facing resource shortages, struggling to meet deadlines, or in need of specialized expertise? Our clinical expert services offer a flexible, tailored solution to meet your short-term or long-term staffing needs—ensuring your clinical study runs smoothly and efficiently and complies to all requirements and regulations.
We provide life sciences companies with the skilled resources needed to execute clinical studies efficiently and compliantly.
From clinical trial associates to medical writers and beyond, our professionals bring the expertise and adaptability required to support your projects—whether it’s short-term, long-term, full-time, or part-time.
We cater to a wide range of needs, including but not limited to:
Managing clinical studies requires the right expertise at the right time. Clinical expert services provides a flexible solution to fill resource gaps without unnecessary recruitment costs.
Here’s why it matters:
Address resourcing needs as they arise, avoiding project delays
No long-term commitments or unnecessary recruitment costs.
Access to skilled clinical research professionals wherever your study takes place.
Our clinical expert services can cover a variety of clinical roles and be customized depending on the exact need in terms of expertise, duration and FTE.
Clinical roles we provide:
Because of our extensive network across the globe, we can also assist you with other types of clinical research-related roles.
.jpg?width=1600&height=900&name=Clinical%20Expert%20Services%20-%20Clinical%20-%20QbD%20Group%20(1).jpg)

We begin with a brief intake conversation to understand your unique needs, allowing us to match you with the right Clinical Research Professional or Subject Matter Expert, considering both expertise and cultural fit.
.jpg?width=540&height=300&name=External%20newsletters%20rectangle%20rechthoek%20(1).jpg)
We share tailored candidate profiles for your review and facilitate conversations with potential candidates to ensure alignment with your team’s needs.
.jpg?width=540&height=300&name=External%20newsletters%20rectangle%20rechthoek%20(2).jpg)
Within the clinical division of QbD Group, we combine extensive clinical research expertise across Medical Device, Biotech, and Pharma sectors with flexible resourcing options tailored to your needs.
Our global network covers Europe and beyond, with diverse expertise in therapeutic areas including but not limited to Cardiovascular, Neurovascular, Orthopedics, and Ophthalmology.
We prioritize continuous development of our employees through an internal training program and maintain the highest standards of quality and regulatory compliance (ICH-GCP and ISO 14155) as part of a global CRO specialized in Medical Devices.
We typically work with an hourly/daily fee which depends on the profile, salary and expertise of the final candidate. Invoices are processed on a monthly basis based on the actual hours spent (in case of an hourly/daily fee).
In case there is a need for a different approach (e.g. fixed fee), this can be discussed as well.
This is no longer an active focus of QbD Clinical.

For several years, we have been working with QbD Group, using their clinical expert services. We appreciate the short communication lines, the seamless fit of contracted profiles with our team, the high quality of their work, and their quick support in resolving any concerns.
We have also recommended QbD Group to our American colleagues and they too have entered into a partnership.











.jpg)


