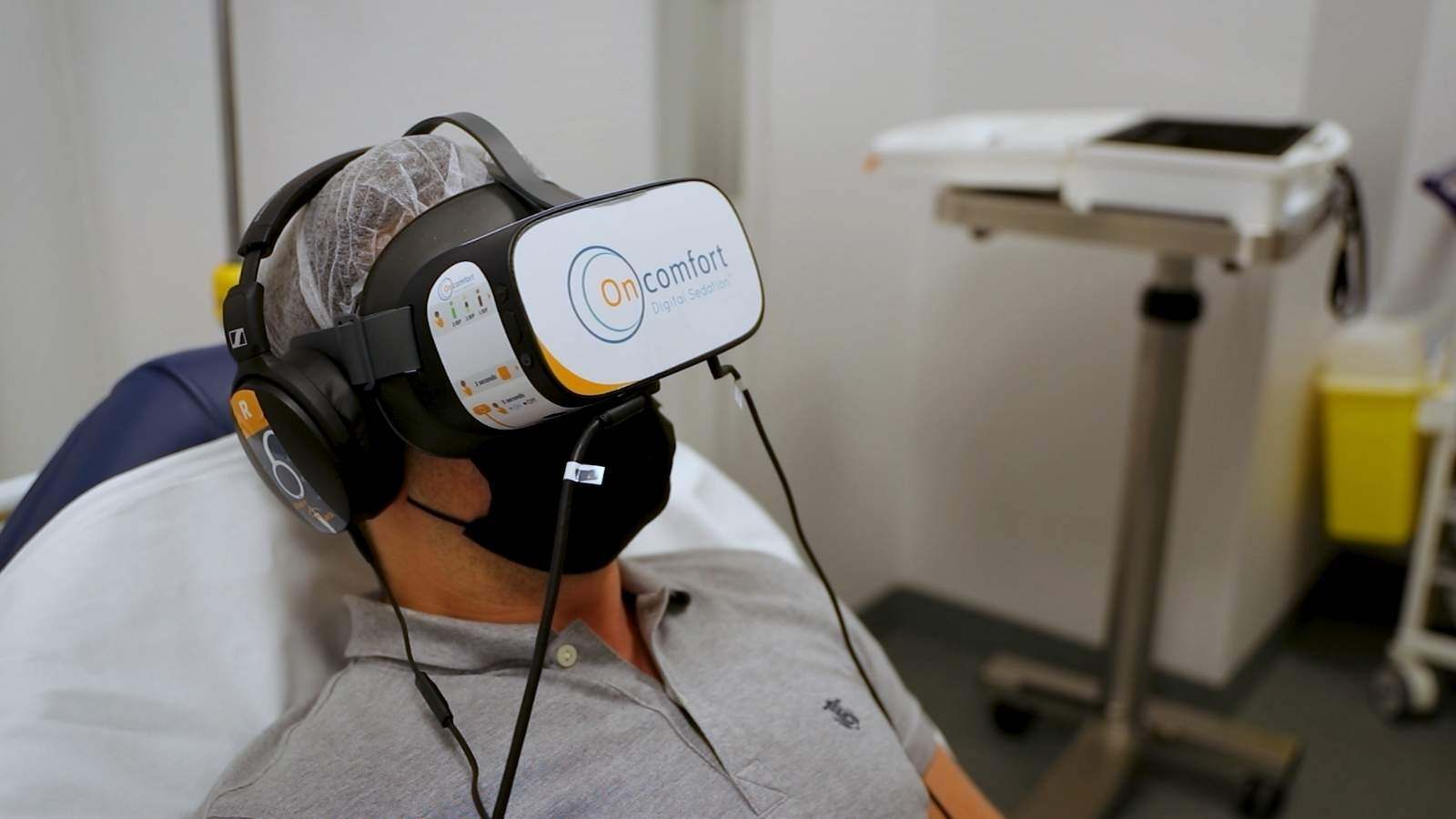
In a previous blog post “Regulatory Inspection Readiness: A Proactive Approach to CAPA and Compliance” we talked about the steps to take after the inspection is over.
But what happens during an inspection…?
With a smile you close the door – following 3 days of intense work, the inspector and his colleague are now done and are leaving your premises. The report can be expected in another 4 weeks.
Looking at the team involved in the inspection, many look relieved but also a bit tired, and maybe even there is a little anxiety. After all, the inspector did have some remarks at the close-out meeting… Could you have been better prepared?
Inspections by Regulatory Authorities are a standard part of the regulatory framework worldwide. It’s likely that, at some point, an inspector may evaluate your company or services.
What to expect during an inspection?
The opening meeting: often an indicator of “points of interest”
In many inspections there is an opening meeting – and when not planned, it is certainly best practice to request one. During this meeting you not only present the best of your company, but the inspector will give intended, or unintended, an indication what are their points of interest for the duration of the inspection.
This means that immediately following the Opening Meeting you may already look at documentation and/or interviewees you had not anticipated or prepared, and get this ready for when the moment during the inspection indeed asks for this information.
“Death by documentation”: a practice to avoid
Inspectors are likely to have requested documents and/or data ahead of the actual visit or contact, but will most often request additional materials in the form of documents or records during the visit.
When you have prepared well, you will easily find the documents and records requested and you can keep documents coming in a good flow.
- Only provide what was asked for.
When not specific, either ask for a specification, or select a “clean / best” recent example - Provide it as soon as it is available.
Even in a situation whereby 5 documents were requested at the same time, if you can provide 2 now, and 3 later, go with the 2 now (unless the inspector indicates differently)Maintaining a good document flow is also an indicator of good document control.
-
Keep track of the request and exactly what was provided.
This will help you in the follow-up of the inspection afterwards (see: ”Regulatory Inspection Readiness: A Proactive Approach to CAPA and Compliance”)
Another tip is:
Keep control over the site visit
Inspectors may request a tour of the facilities. This is fairly standard practice, however you will want to maintain control over this visit. So act like a tour-guide.
- Follow a logical flow of events, and guide the inspector from one area to the next, without leaving much room for
the inspector to wander off or go somewhere else. - Don’t hide anything, and make it obvious that what they’re seeing is relevant, even if it means they will not have seen everything.
If you’ve prepared well in advance, you should not be seeing outdated materials on notice boards, or people chatting about confidential information in the corridors. If it happens, discretely, as if a matter of routine, address the situation without drawing attention to it.
The dreaded interview…
In almost all situations, interviews are routinely part of an inspection, whether it be on site or virtual. All colleagues may be subject to interviews, but usually the inspection agenda (when received upfront) and opening meeting will give an indication who will be interviewed.
Also for interviews, it is possible to prepare.
Interviews are a possibility to shine! This is your chance to demonstrate, explain what you do, and why. You are the owner of your role and responsibilities. Meaning, you are the best person to explain.
Standard questions include information on your training and experience, and your role and responsibilities within the scope of the inspection.
Think about answering any question making use of the 5 W- and 1H elements: WHY – WHEN – WHAT – WHERE – WHO and HOW.
Other tips include
- If you don’t know the answer, say so. Don’t go invent information to try and please the inspector.
- Either you should know, but can’t remember. It is fine to indicate you need to look it up and provide supporting documentation if needed.
- Either you’re not the best person to answer. It is fine to indicate the inspector should raise this question to an appropriate colleague.
- Only answer the question that was asked. Don’t add information to fill in a void or because it looks “good” to talk. This is the time to be obnoxious. Just keep it Simple and Short, respect the truth, but don’t elaborate.
- Remain truthful, even if it may make the company look not perfect…
Daily debrief? Or wait and see?
In multi-day inspections, a Daily Debrief may be foreseen by the inspector. When this is not the case, it is a good idea to request one. This is an opportunity to re-address and/or prepare for the next day.
Inspectors are not infallible. They may have seen and heard certain elements that are an area of concern, but maybe they don’t have the full picture (yet). Picking this up during a debrief, can allow you to re-address the area of concern, or prepare additional documents, records and/or even interviews for the following day.
There is no need to go into lengthy discussions – after all also the inspector is doing their job. When something is insufficient, you already have ammunition to start your solution building early.
The end… closing meeting
At the end of the inspection, the inspector should foresee a summary of what they have seen. When this is not planned, again, it is a good idea to ask for some time together.
This is not a time to go into discussions, it is not a re-visiting of the entire inspection. But there may be an opportunity to rectify certain situations still.
The inspector will not provide graded results. But they will be able to share what they have seen and heard, and if they have concerns about anything, but also will provide feedback on what they are happy about. Usually this feedback will be verbal, so it’s a good idea to have a note taker available. Occasionally the inspector may share an informal written summary as well.
A US Investigator (inspector) may present an FDA Form 483 – which is an indication of important areas of concern.
On many an occasion, the inspector will have seen something that they would prefer to see differently, though not always. Sometimes you just did get it right – at least on the domain the inspector’s reviewed.
The past days (or weeks) have been spent in agony, having the feeling to have to justify yourself (even if it’s just explaining…) and chasing documents, records in your systems, chasing interviewees to be on time in their allotted slot. And despite these efforts, the inspector has seen a shortcoming…
Could I have been better prepared? Could these areas of concerns have been avoided?
The answer is yes. Many, if not all, areas of concern could likely have been prevented.
Note: This approach is also applicable to audits by certifying/notified bodies or significant internal audits (or for service providers: key client audits).







.png?width=109&height=108&name=Pharma%20(2).png)
.png?width=111&height=108&name=Medical%20Devices%20(2).png)
.png?width=84&height=107&name=IVD%20(2).png)







.png)

.jpg)