Ensuring the stability of medicinal products is crucial for maintaining their effectiveness and safety throughout their shelf life. Numerous factors come into play when determining the stability of pharmaceutical products, making it a complex process.
Any changes in the physical appearance or chemical composition of a medicinal product can significantly impact its potency and safety.
In this blog post, we will explore the various factors that influence the stability of medicinal products and the parameters that need to be considered during their storage.
ICH Stability Studies on Pharmaceuticals
Stability of Medicinal Products: 4 Influencing Factors
1. Storage time
As a general rule, medicinal products tend to degrade and deteriorate over time. The longer a product is stored, the greater the likelihood of its degradation. This is an important consideration when determining the shelf life of a medicinal product.
2. Storage conditions
Storage conditions, particularly temperature, relative humidity, and exposure to light, play a significant role in the stability of medicinal products. Higher temperatures and humidity levels accelerate the deterioration of dosage forms.
Exposure to light can also lead to increased decomposition. For photosensitive medicines, it is essential to store them in glass amber bottles and keep them in a dark place to maintain their stability.
3. Type of dosage forms
The chemical stability of a medicinal product is influenced by its dosage form. Liquid dosage forms, such as solutions and semisolid forms containing water, are more prone to degradation compared to solid dosage forms. The presence of water makes these forms more susceptible to hydrolysis and other degradation processes.
4. Container and closure system
The choice of container and closure system has a significant impact on the stability of medicinal products. The moisture content within the packaging plays a critical role in the chemical degradation of pharmaceutical formulations. Pharmaceutical companies carefully select primary packages with suitable moisture-barrier properties to control the effects of humidity.
Impermeable containers allow stability studies to be conducted under controlled or ambient humidity conditions. However, it is important to note that glass vials with rubber stoppers may not be considered impermeable by regulatory authorities.
Therefore, stability testing for products stored in impermeable containers can be performed under any controlled or ambient moisture conditions. It should be noted, however, that glass vials with a rubber stopper are not automatically defined as impermeable by any regulatory agency.
However, ICH Q1 A(R2) requires that water-based products packaged in semi-permeable containers be evaluated for potential water loss in addition to physical, chemical, biological and microbiological stability.
For non-aqueous products, but solvent-based products, other similar methods may be developed and reported. This evaluation can be performed under conditions of low relative humidity, as discussed below.
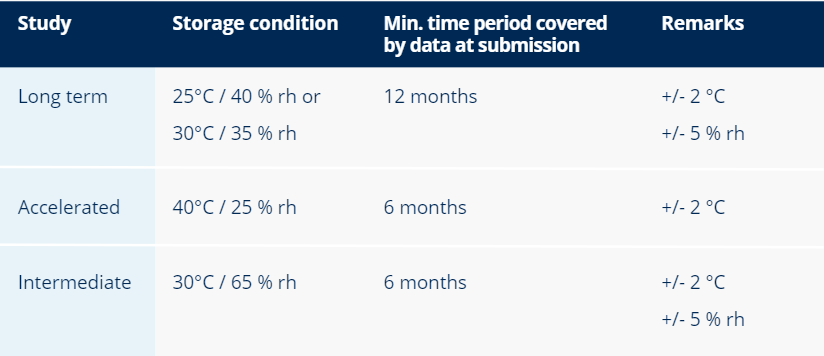
Ultimately, it should be demonstrated that water-based drugs stored in semi-permeable containers can withstand low relative humidity environments. This evaluation can be performed by conducting a stability study under low relative humidity conditions.
The conditions prescribed by the ICH are listed in the table above. An alternative approach is to perform the stability study at a higher relative humidity and derive the water loss at the reference relative humidity by calculations.
Conclusion
The stability of medicinal products is influenced by several factors, including storage time, storage conditions, dosage form, and container and closure systems. Pharmaceutical companies must carefully consider these factors when determining the shelf life of a medicinal product.
Understanding and controlling these variables is essential for maintaining the potency, safety, and effectiveness of pharmaceuticals, ultimately ensuring the well-being of patients who rely on them.
By conducting comprehensive stability studies and following regulatory guidelines, manufacturers can ensure that their medicinal products maintain their stability throughout their intended shelf life.

ICH Stability Studies on Pharmaceuticals
Quercus, your trusted GMP Lab
Quercus Laboratories is a member of the QbD group and is a trusted GMP Laboratory that supports the pharmaceutical industry with a wide range of QC testing services. We offer a complete service from chemical and microbiological QC testing of raw materials and finished drug products.
Our capabilities include:
Analytical Method Validation – Batch release – EU Import services – Tech transfers of analytical methods – Batch Testing and Certificate of Analysis – Manufacturing plant certification for EU-GMP compliance – Documentary review – Batch Certificate for release – QP Back-Up Services – Additional stability studies – Secure storage of retention samples – MA holder – Regulatory affairs support: CTD dossier writing and marketing authorization application (CP/DCP/MRP/NP)

Need reliable lab testing and validation?
About the Author
MP, QP · Division Head Lab Services
Yves leads the Lab Services division at QbD Group, bringing deep expertise in GMP laboratory operations, analytical method validation, and quality control for pharmaceutical manufacturing.
Subscribe to the latest updates in life science
Expert perspectives delivered to your inbox — pick your interests.
No spam, ever. Unsubscribe anytime.